Introduction
Barocaloric (BC) materials are rapidly emerging as leading candidates for enabling commercial solid-state heating and cooling technologies1,2,3,4,5,6,7,8. These materials are characterised by their ability to undergo substantial isothermal entropy changes (\(|\Delta {S}_{{\rm{it}}}|\)) and adiabatic temperature changes (\(\Delta {T}_{{\rm{ad}}}\)) when subjected to hydrostatic pressure. To date, materials exhibiting colossal barocaloric effects (BCEs) (\(|\Delta {S}_{{\rm{it}}}|\) > 100 J K−1 kg−1) have been discovered across various material families, including plastic crystals9,10,11,12,13,14,15,16,17, polymers18,19 and hybrid organic-inorganic materials20,21,22,23. As solid-state alternatives, these materials have negligible global warming potential, offering a stark contrast to vapor refrigerants that risk leakage into the atmosphere24 and can be toxic, flammable and/or greenhouse gases. Furthermore, BC materials have been proposed to achieve coefficients of performance (COP) several times higher than those of existing vapor compression systems25,26. This potential for increased sustainability and efficiency is a key driver of BC materials research.
Despite the discovery of numerous materials exhibiting BCEs, the search for a technologically relevant BC material continues because of a need to balance multiple practical requirements. Viable BC materials must not only match their phase transition temperatures to the desired application but should, ideally, operate reversibly, with minimal hysteresis and within a pressure limit of ~1 kbar16,26. Achieving this without compromising the enthalpy and entropy changes of the phase transition can be challenging, and it is often the case that the headline colossal entropy change promised by a new material cannot be accessed in a reversible and practicable device. Amongst the candidate BC materials, plastic crystals (PCs) are perhaps the most extensively studied, owing to the large entropy changes associated with their order-disorder phase transitions, driven by molecular reorientations. The first colossal BCE was discovered in the PC neopentyl glycol, (CH3)2C(CH2OH)2, (NPG)9,10, and subsequent studies have shown that related materials, such as pentaglycerine, (CH3)C(CH2OH)3, (PG), also display a BCE11. However, these materials suffer from significant reversibility issues arising from supercooling effects27,28 that cause hysteresis in the temperatures at which heat can be rejected and absorbed. Phase transition reversibility can only be achieved at substantially elevated pressures, which is unsuitable for a commercialised device. Additionally, as NPG exhibits a BCE near room temperature, its operational temperature is not ideal for refrigeration purposes7, and the BCE of PG occurs at temperatures unsuitable for most heating and cooling applications.
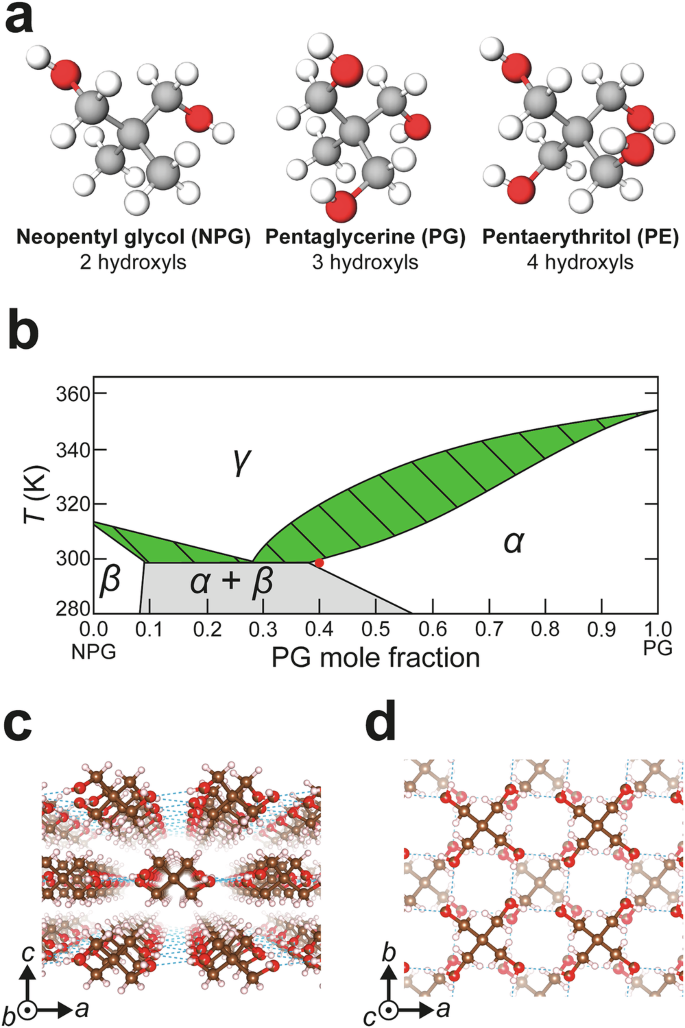
Beyond the context of BC applications, forming solid solutions of the neopentyl plastic crystals NPG, PG and pentaerythritol, C(CH2OH)4, (PE), has been investigated in detail29,30,31,32,33,34. It is well known that the solid-solid (S-S) phase transition temperatures can be tailored through compositional variation. It has also been shown that the doping of pure NPG with organic and inorganic molecules can improve its thermal conductivity35,36,37,38, but without clear improvement of BC performance. The three neopentyl plastic crystal molecules are shown schematically in Fig. 1a, all of which have a tetrahedral structure and differ in the number of hydroxyl units that underpin the hydrogen bonding networks in the ordered crystal (OC) phase. All three PCs undergo S-S phase transitions from a lower entropy OC structure to a higher entropy plastic crystal (PC) structure at temperatures related to the number of intermolecular hydrogen bonds supported by the molecule (~314 K for NPG, ~354 K for PG and ~460 K for PE). Both PG and PE adopt the same OC structure, body-centred tetragonal (BCT, I\(\bar{4}\)), with hydrogen bonding stabilising a continuous layered structure (Fig. 1c). NPG instead adopts a monoclinic (\(P{2}_{1}/c\)) structure32, stabilised by intermolecular hydrogen bonds between chains of molecules, resulting in a more needle-like crystal habit28. Upon heating, all three materials adopt a face-centred cubic (FCC, \({Fm}\bar{3}m\)) structure in the PC phase.
a Molecular schematics of neopentyl plastic crystals, each tetrahedral molecule differing only in the number of hydroxyl functional groups. b NPG-PG binary phase diagram adapted from calculated data in the literature32,33. \(\alpha\), \(\beta\), and \(\gamma\) correspond to BCT, monoclinic and FCC crystal structures, respectively. The grey section indicates the compositions where the ordered crystal phases are immiscible. The green hatched section indicates the phase coexistence region during ordered crystal to plastic crystal phase transitions. The red dot shows the target binary composition in this work. c,d) Structural schematics of the BCT structure from two orthogonal perspectives. Hydrogen bonds form layers of molecules in the \({ab}\) plane with van der Waals forces between layers along the \(c\) direction. Schematics were drawn using VESTA64.
For practicable BC refrigeration, phase transitions cooler than that of NPG (314 K) and with minimal thermal hysteresis are desirable6,7. Furthermore, the solid solutions should ideally transition between two well-defined structures, without phase segregation and decomposition concerns, to avoid so-called ‘continuous’ phase transitions that can span a temperature range of up to 100 K33. Figure 1b shows a schematic of the NPG-PG binary phase diagram reproduced from data calculated elsewhere32,33. Here, \(\alpha\), \(\beta\), and \(\gamma\) indicate the BCT, monoclinic and FCC structures, respectively, adopted by the NPG-PG binary solid solution. This phase diagram indicates that the lowest transition temperature of ~300 K (under ambient pressure) that can be reached for a single-phase NPG-PG binary solid solution is for molar compositions of either 8% NPG:92% PG or 60% NPG:40% PG (the latter marked with a red dot in Fig. 1b). Previous calorimetric studies of NPG-PG solid solutions have shown that both compositions exhibit significant thermal hysteresis upon cycling29 and so are not suitable for practicable devices.
Our hypothesis is that organic dopants could be used to disrupt the hydrogen bonding network that underpins the phase transition, reducing supercooling effects and the problematic thermal hysteresis. We chose small quantities of PE as a dopant, exploiting its similar molecular size and structure but different number of hydroxyl units to the NPG molecule. The 60% NPG:40% PG solid solution is also isostructural with PE, which makes it likely that a single-phase ternary solid solution of the three molecules can be formed. The expected BCT phase, \(\alpha\), is characterised by an extensive intermolecular hydrogen bonding network in the \({ab}\) plane and relatively weaker van der Waals interactions along the \(c\)-direction39, as shown in Fig. 1c, d. Our strategy is to introduce different neopentyl molecules into this structure to modify the hydrogen bonding landscape due to the differing numbers of hydroxyl functional groups, ultimately affecting the phase transition conditions for the solid solution. Although binary and ternary solid solutions of the neopentyl PCs have been studied extensively, their BC performance has not been investigated. Furthermore, all our measurements were carried out over a period of approximately 15 months, demonstrating the long-term thermodynamic stability of the solid solutions. This strategy has the potential to optimise BC properties through compositional engineering in neopentyl PCs and other BC molecular systems, such as functionalised adamantane12,14,40,41,42, which, unlike pure adamantane43, have transition temperatures close to room temperature.
We present results for a binary solid solution of 60% NPG and 40% PG in comparison to the same composition doped with a small amount of PE, i.e. a ternary solid solution of 60% NPG, 38% PG, and 2% PE, and benchmark these materials against pure NPG. Both solutions retain the colossal barocaloric effect observed in NPG, and the main result is that we are able to access a greater fraction of that colossal entropy change under reversible, isothermal conditions with pressures \(\le\)1 kbar. In comparison to NPG, the ternary solid solution has a seven times greater reversible isothermal entropy change (\(\left|\Delta {S}_{{\rm{it}},{\rm{rev}}}\right|\) = 13.4 J kg−1 K−1) and its operational temperature span (\(\Delta {T}_{{\rm{span}}}\) = 18 K) is increased by a factor of 20 at the technological pressure threshold of 1 kbar, corresponding to a more than 70 times increase in reversible refrigeration capacity at this pressure. Critically (and in line with previous studies), we can tune the transition temperature of the solid solution to be closer to the operational requirements of cooling applications. To understand these enhancements, we use powder synchrotron x-ray diffraction and quasielastic neutron scattering experiments, which reveal structural and dynamical changes, aiding our understanding of how disrupting the hydrogen bond network can improve the reversibility of the OC → PC phase transition. Neutron spectroscopy fixed window scan techniques reveal that the activation energies for the rotational modes underpinning the BCE are reduced by up to 50% compared to NPG. We propose that the improved reversibility and operational temperature span are facilitated by dopant-induced heterogeneous nucleation sites and a large (\(\alpha\),\(\gamma\)) phase co-existence region spanning around 30 K during thermal cycling. These properties arise from a disruption to the hydrogen bond network caused by substitutional doping. Our findings pave the way for rational design in creating new BC PCs, accessing significant compositional phase spaces and enabling tailored BC performance.
Results
Comparison of barocaloric effects using variable-pressure calorimetry
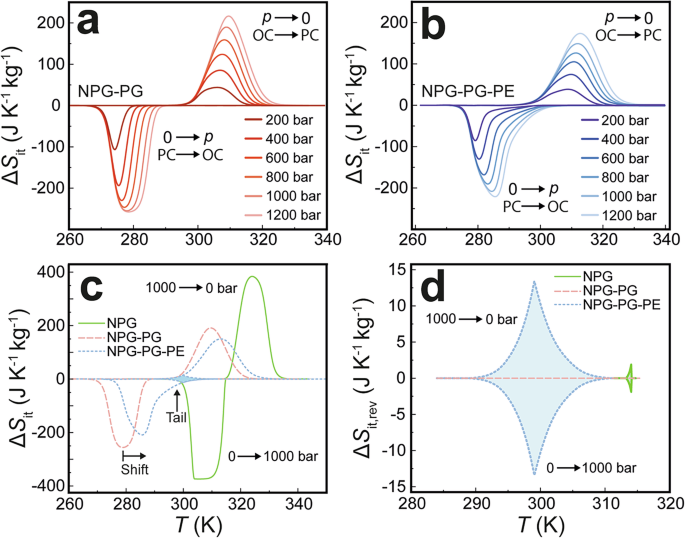
Full characterisation was only conducted on the most promising compositions. We therefore focused on the binary NPG:PG composition that was predicted to have the lowest isothermal phase transition temperature under applied pressure, and that was also expected to transition between two well-defined phases (see Fig. 1): the binary solid solution of 60% NPG and 40% PG (herein denoted as NPG-PG). Ambient-pressure calorimetry (Supplementary Fig. 1) was used to determine the optimal ternary composition, a solid solution of 60% NPG, 38% PG, and 2% PE (herein denoted as NPG-PG-PE). Calorimetric measurements under variable applied pressure were then conducted on both solutions in comparison with NPG, as shown in Fig. 2. In each case, the latent heat is attributed to the reversible OC → PC phase transition. The comparison is evaluated quasi-directly from isobaric calorimetric peaks (Supplementary Fig. 2a, b), using calculations outlined in the Methods Section. This standard method for determining reversible BC entropy changes11,44 allows a conversion from data collected during isobaric thermal cycling to approximate data relevant to pressure cycling. The former is experimentally efficient whilst the latter represents the operation of a realistic BC device. In these plots, the OC → PC (PC → OC) phase transition corresponds to the peaks with positive(negative) \(\Delta {S}_{{\rm{it}}}\) that would be driven by decompression(compression) sweeps in a real device. In each case, the maximum value of \(\Delta {S}_{{\rm{it}}}\) is achieved at the corresponding peak temperature, and so our aim was to minimise the hysteresis. This would bring the peaks into alignment for compression and decompression steps and thereby maximise the absolute reversible entropy change (\(\left|\Delta {S}_{{\rm{it}},{\rm{rev}}}\right|\)) at a fixed temperature.
Isothermal entropy changes (ΔSit) driven by applying (0 → \(p\)) and removing (\(p\) → 0) pressure in a NPG-PG and b NPG-PG-PE. c Comparison of entropy changes between pure NPG, NPG-PG-PE and NPG-PG when driven by \(|\Delta {p|}\) = 1000 bar. d Comparison of the reversible entropy change regime between NPG, NPG-PG and NPG-PG-PE when driven by \(|\Delta {p|}\) = 1000 bar.
A comparison of Fig. 2a (NPG-PG) with Fig. 2b (NPG-PG-PE) reveals that both materials exhibit ‘colossal’ BCEs with \(|\Delta {S}_{{\rm{it}}}|\) ~ 200 J K−1 kg−1. Critically, NPG-PG-PE displays significantly reduced hysteresis in the first-order phase transition, resulting in an enhancement of its reversible BC performance compared to NPG-PG. In contrast, NPG-PG, shown in Fig. 2a, has no reversible entropy change at any of the pressures measured here (\(\left|\Delta {S}_{{\rm{it}},{\rm{rev}}}\right|\) = 0), as the peaks for PC → OC and OC → PC do not overlap at any temperature. Similarly, the reversible entropy change for NPG is known to be small when driven with pressures around 1 kbar9,10. In both cases, the implication is that a compression/decompression cycle under the isothermal conditions of a BC refrigerator based on NPG or NPG-PG could not access both the OC → PC and PC → OC phase transitions when operated with 1 kbar.
NPG-PG-PE has a large region of reversible entropy change, with overlapping PC → OC and OC → PC curves around 300 K. Although the OC → PC peak phase transition temperature is nearly identical for both materials at all applied pressures, the improvement arises because the supercooled PC → OC transition occurs at higher temperatures for NPG-PG-PE (Fig. 2b) and has a distinct asymmetric character. Furthermore, both OC → PC and PC → OC phase transitions are broader for NPG-PG-PE, and are especially pronounced during the supercooled PC → OC transition, when compared to NPG-PG. In Fig. 2c, a comparison of NPG, NPG-PG and NPG-PG-PE with an applied pressure of \(|\Delta {p|}\) = 1000 bar shows that NPG-PG-PE exhibits the largest reversible temperature span (\(\Delta {T}_{{\rm{span}}}\)), as indicated by the shaded blue region. NPG-PG has no reversible isothermal entropy change at this pressure (\(\left|\Delta {S}_{{\rm{it}},{\rm{rev}}}\right|\) = 0, \(\Delta {T}_{{\rm{span}}}\) = 0), highlighting the significant improvement of the BC performance from the addition of just 2% PE to the binary solid solution. Figure 2d demonstrates that at the critical technological threshold pressure of \(|\Delta {p|}\) = 1000 bar, NPG-PG-PE produces a maximum reversible isothermal entropy change of up to \(\left|\Delta {S}_{{\rm{it}},{\rm{rev}}}\right|\) ~13.4 J K−1 kg−1 over a temperature span of \(\Delta {T}_{{\rm{span}}}\) ~ 18.0 K. In comparison, pure NPG under the same pressure change produces a ~ 90% lower reversible isothermal entropy change of \(\left|\Delta {S}_{{\rm{it}},{\rm{rev}}}\right|\) ~1.9 J K−1 kg−1, with a narrow temperature span of \(\Delta {T}_{{\rm{span}}}\) ~ 0.9 K at a temperature of ~314 K. This corresponds to a reversible refrigerant capacity (\({{\rm{RC}}}_{{\rm{rev}}}\)) of 73 J kg−1 for NPG-PG-PE and 1 J kg−1 for NPG at 1 kbar (see Supplementary Fig. 2d for \({{\rm{RC}}}_{{\rm{rev}}}\) values at other pressures). This improvement of BC performance at 1 kbar highlights the capabilities of NPG-PG-PE relative to pure NPG. Further examination of Fig. 2b, c reveals that the broader regime of reversible \(|\Delta {S}_{{\rm{it}}}|\) in NPG-PG-PE results from the increase in asymmetry of the PC → OC phase transition as compared to NPG-PG. The asymmetry in the phase transitions is marked by an extended tail around 300 K in Fig. 2c, clearly contributing to enhanced reversible BC performance. Notably, while the hysteresis in NPG-PG-PE is reduced, the sensitivity of the transition temperature to pressure (\(\frac{d{T}_{0}}{{dp}}\)) remains similar in both NPG-PG and NPG-PG-PE (Supplementary Figs. 3 and 4).
Nucleation density from infra-red imaging
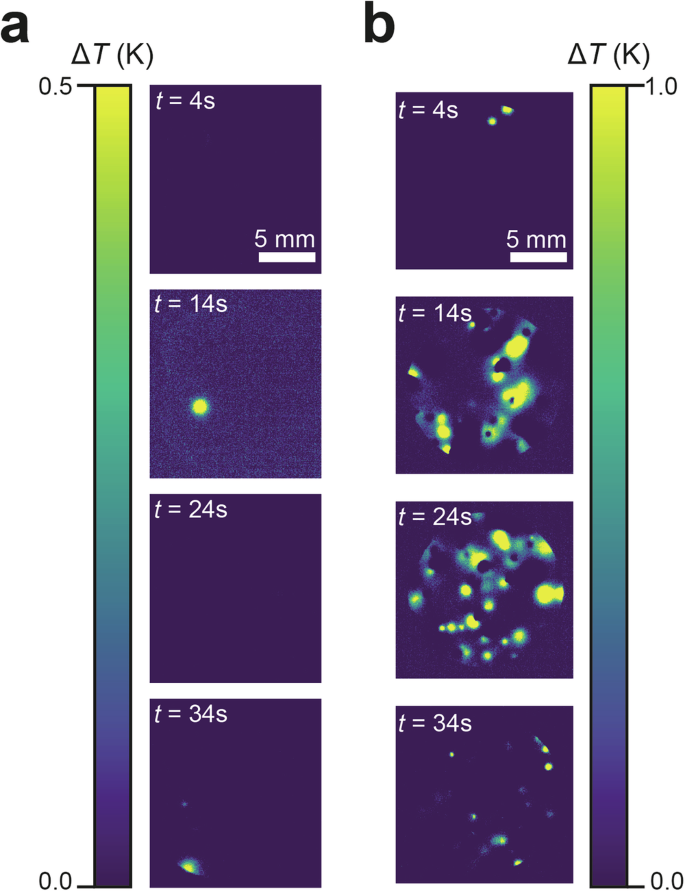
Infra-red (IR) imaging of BC samples provides invaluable spatially-resolved insight into heat emission and absorption, complementing the calorimetry analysis. We imaged all three samples during cooling using an IR camera (Supplementary Figs. 5–7), with the main observations summarised in Fig. 3. As the samples transition from PC to OC phases, the heat released is observed as a local increase in temperature that is readily observed in differences between successive frames of a sequence of infrared images. An advantage of this approach is that it highlights just material that is undergoing the phase transition, either at a nucleation point or at a propagation front; another advantage is that it is more directly relatable to the differential calorimetry measurements discussed above.
Infra-red difference images frames of a NPG-PG and b NPG-PG-PE on cooling through the PC → OC phase transition. Bright regions indicate nucleation events and phase transition propagation. Cooling rate was 5 K min−1. Samples are 13 mm diameter, 0.8 mm thick circular disks, scale bar is 5 mm.
Pure NPG (Supplementary Fig. 5) shows a small number of nucleation events followed by the rapid propagation of phase transition wave fronts through the material, as observed previously28,45. For NPG, the phase transition is clearly linked to a crystallisation process, with the phase transition advancing along needle-like domains. Neither NPG-PG-PE nor NPG-PG (Fig. 3 and Supplementary Figs. 6 and 7) demonstrates the same behaviour. As they become super-cooled, both samples are characterised by a greater number of more localised nucleation events that appear and are quenched without significant propagation of transition fronts through the material. There is no such directionality to the propagation, which suggests a more polycrystalline, isotropic underlying structure with smaller grains and a greater mosaic spread. The direct comparison between NPG-PG and NPG-PG-PE seen in Fig. 3 suggests that the addition of PE dramatically increases the nucleation density in NPG-PG-PE compared to NPG-PG. This larger number of more discrete nucleation events may explain the protracted nature of the PC → OC phase transition measured in calorimetry, which will be discussed further below. Videos showing the IR imaging through the phase transition for each sample can be found in Supplementary Movies 1–3.
Phase transition-induced structural changes
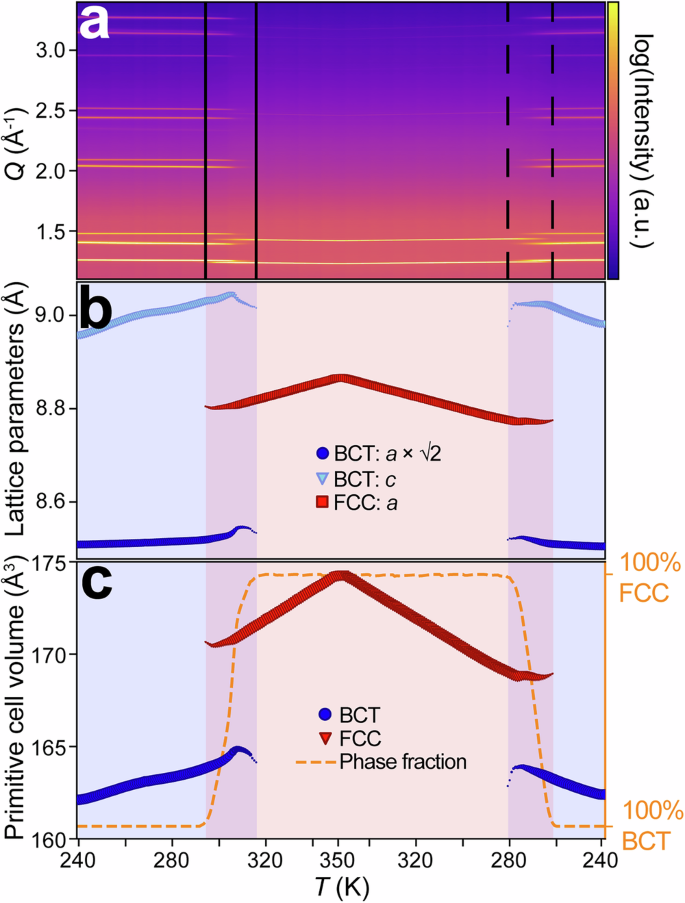
Previous studies have shown that solid solutions of neopentyl-based PCs exhibit a variety of crystal structures33,34,39. For the composition indicated by the red dot in Fig. 1b, we expected NPG-PG to adopt the same OC BCT structure as PG30,33, which has an extensive hydrogen bond network in the \({ab}\) plane and van der Waals interactions between layers in the \(c\)-direction (Fig. 1c, d). The lattice symmetry of NPG-PG-PE, NPG-PG and PG was compared using powder synchrotron X-ray diffraction (XRD) during heating and cooling under ambient pressure (Supplementary Figs. 8–10). Analysis of the NPG-PG-PE structure is presented in Fig. 4, with similar datasets for the other two samples presented in Supplementary Figs. 11 and 12. (Note that comparison with NPG was not included as it adopts a different, monoclinic structure in its low temperature phase.) All three samples show a thermally-activated expansion from a single-phase BCT structure to a single-phase FCC structure: we confirm that NPG-PG adopts the structure predicted by Fig. 1b and also confirm that the addition of 2% of PE does not alter that crystal symmetry. The low temperature structure, including molecular conformations, was confirmed using single-crystal electron diffraction (Supplementary Information Note 1) for both PG and NPG-PG-PE at 150 K, which verified that they are isostructural, with varying occupation of hydroxymethyl and methyl groups in accordance with their composition.
a Full powder XRD data for NPG-PG-PE on heating and cooling. b Thermal variation of the refined lattice parameters throughout a heating and cooling cycle. BCT lattice parameters at 246 K: \(a\) = 6.0170 Å, \(c\) = 8.9642 Å. FCC lattice parameters at 319 K \(a\) = 8.8234 Å. To facilitate a direct comparison between related vectors, the BCT \(a\)-parameter is multiplied by a factor of \(\surd 2\). c The primitive unit cell volume determined from the lattice parameters. The dashed orange line shows the FCC phase fraction as a function of temperature, with the shaded red region indicating the phase transition region on heating and cooling. This clearly demonstrates the extended phase coexistence region from below 300 K to nearly 320 K on heating.
A comparison of the refined lattice parameters (also plotted in Fig. 5a) shows that NPG-PG-PE is ~0.4% contracted in the \({ab}\) plane and ~1.2% expanded along the \(c\)-axis compared to PG. A similar trend is observed in comparing NPG-PG with PG, and there is a slight expansion of the \({ab}\) lattice with the addition of PE to form NPG-PG-PE. Such subtle anisotropic changes arise from a combination of steric effects and the potential for additional hydrogen bonding. Addition of hydroxyls will increase the physical size of molecules, potentially expanding the surrounding lattice and consequently reducing the strength of hydrogen bonding. On the other hand, if the hydroxyls are oriented favourably, then increased hydrogen bonding may reduce the local intermolecular separation. Although the hydroxyls can orient to lie within the ab plane, recent simulations also suggest that the degree of hydrogen bonding may be less than expected on the basis the hydroxyl count46. In the present case, expansions in the \({ab}\) plane with increased hydroxylation suggest that steric effects and hydroxyl orientation are important.
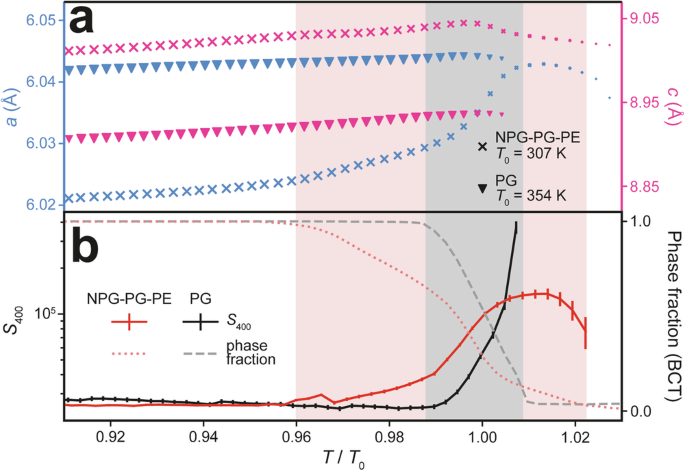
a NPG-PG-PE and PG BCT lattice parameters as a function of reduced temperature, \(\frac{T}{{T}_{0}}\), showing \(a\) (blue, left axis) and \(c\) (pink, right axis) as samples are heated through their phase transitions. The relative sizes of the markers encode the phase fraction. b Anisotropic strain parameter \({S}_{400}\) and phase fraction as a function of \(\frac{T}{{T}_{0}}\). The data scale for PG has been increased by a factor of four. Red and grey shaded sections indicate the phase coexistence regions for NPG-PG-PE and PG, respectively.
For all three samples, individual phases show approximately linear thermal expansion away from the phase transitions. The OC → PC phase transition is then characterised by a substantial increase in intermolecular separation in the \({ab}\) plane and corresponding reduction of the intermolecular spacing along the \(c\)-axis (Fig. 4a, b), producing an overall volumetric expansion (Fig. 4c). Overall, a volume change of ΔV = 4.8% (Fig. 4c) occurs through the phase transition in NPG-PG-PE, obtained from the refinements (Supplementary Fig. 10) at 292 K (BCT) and 319 K (FCC). It is notable that this volume change is close to that of pure NPG11. These observations are consistent with the loss of hydrogen bonding in the \({ab}\) plane as the molecules start to rotate, transitioning to an FCC structure with non-directional bonding. The equilibrium phase transition temperatures (on heating, \({T}_{0}\)) are \({T}_{0}\) = 307 K for NPG-PG-PE, \({T}_{0}\) = 302 K for NPG-PG and \({T}_{0}\) = 354 K for PG, which compare well with the ambient pressure calorimetry data (Supplementary Fig. 1) and demonstrate an ability to reduce the transition temperature of PG through appropriate blending and doping.
Closer analysis of XRD data reveals that the NPG-PG-PE phase transition occurs over a wider temperature range than that of isostructural PG. Figure 5a compares the temperature dependence of the BCT lattice parameters for NPG-PG-PE and PG after four initial thermal cycles (lattice parameters shown in Supplementary Fig. 13) as they are heated towards their transition temperatures (NPG-PG is shown in Supplementary Fig. 14 to behave similarly to NPG-PG-PE). The shaded areas indicate the temperature ranges where both OC and PC phases co-exist, determined from the refined phase fraction in Fig. 5b. For both NPG-PG (Supplementary Fig. 11) and NPG-PG-PE, the \(\alpha\) and \(\gamma\) phases coexist across a range of ~30 K (pink shading), substantially wider than the ~8 K span for PG (grey shading). For \(\frac{T}{{T}_{0}}\) < 0.96, the temperature dependence of the unit cell parameters of the BCT phase in all materials is dominated by the roughly linear trends expected for thermal expansion. The main contribution to the volume expansion of the BCT phase is the relatively large changes in \(c\) (note the scales in Fig. 5a), presumably due to the weaker van der Waals bonding in this direction. Approaching the transition (\(\frac{T}{{T}_{0}}\) > 0.96), there is a notable, non-linear increase in the expansion of the NPG-PG-PE \(a\) lattice parameter (blue, left axis). This trend is not observed in PG, where a linear trend continues up to the transition. The non-linear behaviour in both NPG-PG and NPG-PG-PE (compared with PG in Supplementary Fig. 14) systems suggests disruption of the hydrogen bond network beyond simple thermal expansion as \(\frac{T}{{T}_{0}}\) = 1 is approached.
A similar anisotropy is observed in the breadth of \({hkl}\) diffraction peaks close to the transition temperature. We fitted Stephens’ anisotropic broadening parameters47, \({S}_{{hkl}}\), to \({hkl}\) reflections from each sample (see Methods Section and Supplementary Fig. 15). Figure 5b compares the temperature dependence of \({S}_{400}\) for PG and NPG-PG-PE, showing that \(h00\) type reflections begin to broaden in NPG-PG (Supplementary Fig. 15) and NPG-PG-PE systems for \(\frac{T}{{T}_{0}}\) > 0.96. In contrast, increased broadening of the PG \(h00\) type reflections is only observed at the start of the full phase transition around \(\frac{T}{{T}_{0}}\) ~1.00. \({S}_{004}\) fits to the broadening of \(00l\) type reflections show relatively smaller increases compared to \({S}_{400}\) at the phase transition for all materials, whilst \({S}_{220}\) type broadening is only required to fit the data for NPG-PG and NPG-PG-PE (Supplementary Fig. 15). These observations again highlight a clear anisotropy in the thermal expansion of the PCs close to their phase transition, and also clear differences between PG and the blends NPG-PG-PE and NPG-PG. Around \(\frac{T}{{T}_{0}}\) ~ 1.00, the first-order region of the phase transition in all samples is characterised by a rapid increase in \({S}_{400}\) that correlates with a rapid change in the phase fraction. However, the more gradual variations in lattice parameter and peak broadening observed for both solid solutions suggest a weakening and perhaps localised disruption of the hydrogen bond network that define what we term the weakly first-order region of the transition. Similar protracted phase transitions are observed upon cooling from the PC back to the OC phase. Supplementary Fig. 16 includes the \({S}_{{hkl}}\) parameters on cooling for all three samples, where NPG-PG-PE has significantly enhanced peak broadening compared to the other two materials, suggesting a greater degree of structural disorder for this sample. As discussed below, we propose that this increased disorder, particularly in the supercooled state, is intricately linked to the increased nucleation events observed by IR imaging during the supercooled transition in NPG-PG-PE.
Molecular dynamics from quasielastic Neutron Scattering
Neutron scattering techniques allow dynamical changes in NPG-PG-PE to be correlated with the observed thermal and structural behaviour, as previously shown for other BC PCs10,43,48,49,50. Quasielastic neutron scattering (QENS) probes the molecular dynamics of hydrogen-containing materials by measuring low energy transfers, typically ranging from meV to µeV, which correspond to dynamic timescales of picoseconds to nanoseconds. QENS datasets contain intensity as a function of energy (\(E\)) transfer and momentum (\(Q\)) transfer (\(S(Q,E)\)) and feature an elastic peak (centred at \(E\) = 0) that is broadened by quasielastic scattering caused by rotational and translational diffusion processes within the sample51. Thus, the degree of broadening is directly correlated with the dynamics occurring within the material. In recent work, we used QENS to identify the key molecular rotational modes responsible for the BCE in NPG and employed inelastic fixed-window scans (FWS) to directly observe a hysteresis in the molecular dynamics50. Here, we again used the IN16B spectrometer at the Institut Laue-Langevin (ILL), which uniquely enables such inelastic FWS measurements by capturing the quasielastic signal within specific narrow energy ranges. Importantly, this technique allows molecular dynamics to be tracked whilst the sample undergoes thermal cycling, providing direct comparison with diffraction and calorimetry measurements. Given the structural similarity between NPG, PE and PG, differing only in the ratio of hydroxymethyl to methyl groups, we apply similar QENS/FWS data analysis to that used for NPG to understand the molecular dynamics in NPG-PG-PE. Given that the binary NPG-PG did not exhibit a reversible BC effect at 1.2 kbar or below, we did not perform QENS analysis on this material. Details regarding the QENS/FWS data acquisition and analysis can be found in the Methods Section and Supplementary Information Note 3.
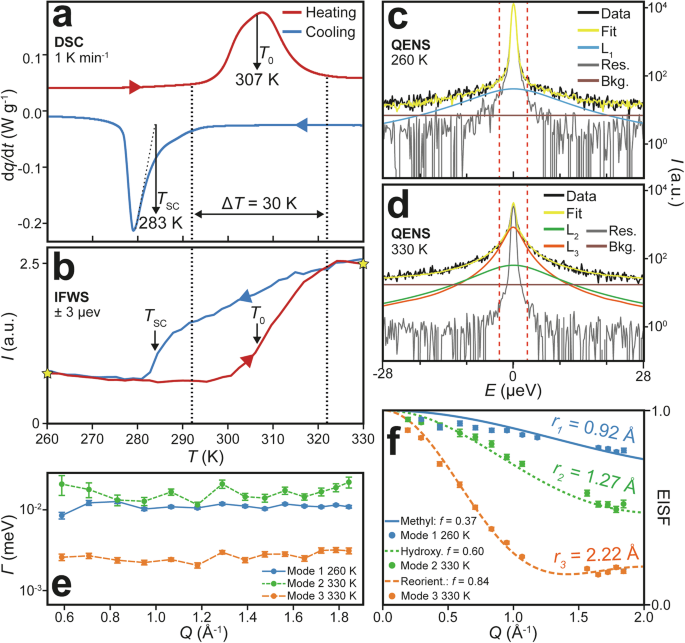
Figure 6a shows a thermal cycle for NPG-PG-PE using ambient pressure calorimetry with a scanning rate of 1 K min−1. Thermal hysteresis is observed in the difference between the heating and cooling peaks, and the data in Fig. 2b show that the peak overlap, and hence the accessible latent heat under isothermal conditions, increases with pressure. In Fig. 6a, there is a temperature range of approximately 10 K where the evolution of latent heat is reversible, even at ambient pressure, which is atypical for PCs. An ambient pressure equilibrium phase transition temperature of \({T}_{0}\) = 307 K, in good agreement with the XRD data, is determined from the peak temperature on the heating step and a supercooled phase transition temperature of \({T}_{{\rm{SC}}}\) = 283 K is obtained from the intersection of the cooling peak inflection point gradient with the temperature axis. Both temperatures are indicated in Fig. 6a with black arrows. The onset is used for the cooling transition due to significant asymmetry, characteristic of supercooled phase transitions. The large phase coexistence region obtained from the XRD phase fraction data on heating is indicated by the double-headed arrow in Fig. 6a, demonstrating that calorimetry and XRD measurements are well correlated. The \(\Delta T\) of 30 K, shown in both techniques, is an order of magnitude greater than that of NPG obtained at the same ramp rate28. This implies a gradual, broadened first-order phase transition where both phases coexist over a broad temperature range. It also indicates that domain kinetics may play an important role in the phase transitions of NPG-PG-PE. The calorimetry data can be directly correlated with inelastic FWS (IFWS) measurements, also obtained at ambient pressure and a heating rate of 1 K min−1, shown in Fig. 6b. For IFWS measurements, rather than measuring quasielastic scattering across all energies at a constant temperature, as is usual in QENS (\(S(Q,E)\)), instead a specific energy window is chosen to measure quasielastic scattering as a function of temperature (\(S(Q,T)\)). This makes it possible to infer contributions from modes in that energy window and to track their temperature dependence. A larger intensity in the IFWS plot implies increased quasielastic scattering within the chosen energy windows, in this case located at \(\pm\) 3 μeV. Thus, a sudden increase(decrease) in IFWS intensity on heating(cooling), as indicated by \({T}_{0}\)(\({T}_{{\rm{SC}}}\)) in Fig. 6b, corresponds to increased(decreased) molecular dynamics in the sample. This is expected when observing the liberation(freezing) of the PC modes at \({T}_{0}\)\(({T}_{{\rm{SC}}})\). Thermal hysteresis in the molecular dynamics is observed as a retention of the quasielastic scattering signal to lower temperatures. The parameters \({T}_{0}\), \({T}_{{\rm{SC}}}\) and \(\Delta T\) obtained from ambient pressure calorimetry and XRD are well aligned with the features observed in the IFWS data, indicating that it is possible to correlate the thermal, structural and dynamical information of NPG-PG-PE.
a Ambient pressure calorimetry data showing the latent heat peaks associated with the phase transition on heating (\({T}_{0}\)) and cooling (\({T}_{{\rm{SC}}}\)). The region between the vertical dashed lines highlights the temperature span of the phase transition during heating, as determined from the XRD phase coexistence region. Note: We intentionally use lowercase \(q\) to represent heat to avoid confusion with the scattering variable \(Q\). b Quasielastic neutron IFWS measurements on heating and cooling with two 0.75 μeV wide energy windows at \(\pm\)3 μeV, collecting the momentum transfer range 0.43 Å−1 < \(Q\) < 1.84 Å−1. Yellow stars indicate temperatures at which QENS scans were obtained. Representative QENS fits for \(Q\) = 0.83 Å−1 at (c) 260 K and (d) 330 K. The red vertical dashed lines correspond to the IFWS measurements at \(\pm\)3 μeV. e Mode linewidths (\(\varGamma\)) as a function of \(Q\) for each of the observed modes in the QENS data. f \({\rm{EISF}}(Q)\) plot for the three identified modes with fitting to rotational geometric models representing methyl, hydroxymethyl and molecular rotations. Data between 1.1 Å−1 < \(Q\) < 1.5 Å−1 were masked due to the presence of structural Bragg peaks in this \(Q\)-range.
To characterise the dynamic modes, full QENS scans were obtained on heating by isothermally measuring at 200 K, 230 K, 260 K, 330 K and 360 K with the scans either side of \({T}_{0}\) shown by the yellow stars in Fig. 6b. The fitting of QENS scans at 260 K and 330 K for \(Q\) = 0.83 Å−1, representative of other \(Q\) values (Supplementary Fig. 19), can be seen in Fig. 6c, d where the red dashed lines indicate the energy windows (\(\pm\)3 µeV) at which the IFWS in Fig. 6b were obtained. The fitting was to the global dataset consisting of both QENS and IFWS, rather than for individual spectra, and was constrained using the IFWS data across all \(Q\) values (Supplementary Information Note 3). Within this model, each Lorentzian (\({L}_{{\rm{i}}}\)) represents a dynamical mode detected within the sample, with a characteristic amplitude and linewidth frequency. The full width at half maximum (FWHM) linewidth of the fitted Lorentzians (\({L}_{{\rm{i}}}\)) as a function of \(Q\), \(\varGamma (Q)\), is shown in Fig. 6e. Here, the \(Q\)-independent behaviour indicates localised rotational dynamics rather than long-range diffusion processes. As was also observed in pure NPG previously, we observe one mode below the phase transition (260 K) and two additional modes in the PC phase above 330 K. We assign these modes as \({L}_{1}\) = methyl rotation with radius \({r}_{1}\), \({L}_{2}\) = hydroxymethyl rotation with radius \({r}_{2}\) and \({L}_{3}\) = molecular reorientation with radius \({r}_{3}\), through calculating their elastic incoherent structure factors (\({\rm{EISF}}(Q)\), Fig. 6f) and fitting the data to known geometric models with good agreement (see Supplementary Information Note 3 and Supplementary Fig. 20 for the geometric origins of \({r}_{{\rm{i}}}\)). The relative magnitudes of the fitted radii are consistent with the modes that they represent, i.e. \({r}_{1}\) (methyl rotation) <\({r}_{2}\) (hydroxymethyl rotation) <\({r}_{3}\) (molecular reorientation). We included a fractional fitting parameter (\(f\)) to account for scatterers that do not participate in the respective process49,50,52,53,54. For Mode 1, the methyl rotation radius was fixed to the value obtained from crystallography55, and we find that \(f\) is lower than it is for pure NPG50, which is consistent with NPG-PG-PE having ~ 20% fewer methyl groups. In fact, for NPG-PG-PE the fraction of 1H atoms in methyl and hydroxymethyl groups is 0.395 and 0.605, respectively, agreeing very well with the fitted \(f\) parameters of 0.37 for Mode 1 and 0.60 for Mode 2. The fit for Mode 3, molecular reorientation, compares well with that obtained for pure NPG50, where a fractional fitting parameter \(f\) < 1.0 is also observed. Since all 1H atoms are expected to be involved in this rotational motion, \(f\) should be unity, and a possible explanation is that not all orientations are accessible at the phase transition temperature48,50,56. Together, these fits indicate that the rotational modes present in NPG-PG-PE are very similar to those of pure NPG, which is to be expected given the chemical similarity of the neopentyl plastic crystals.
We now compare features in the calorimetry data in Fig. 6a with those in the IFWSs in Fig. 6b. On heating, the increase in IFWS signal matches the endothermic latent heat peak in calorimetry, with the inflection point of the IFWS signal approximately coinciding with the peak latent heat temperature. These observations correspond to the OC → PC phase transition. Since the IFWS signal increases during this transition, it is evident that additional dynamical modes are activated on heating. On cooling, the calorimetry data appear to show two-component behaviour in the exothermic latent heat peak. Firstly, the latent heat peak slowly increases from 300 K to 288 K, indicating the weakly first-order region, followed by a sharp increase centred around 280 K, characteristic of a supercooled phase transition. The slow decrease of IFWS intensity with cooling indicates (1) a gradual slowing of molecular dynamics and/or (2) a reduction in the number of scatterers exhibiting the PC molecular dynamics. This weakly first-order region is followed by a sharp decrease in the IFWS intensity, coinciding with the sharp exothermic latent heat peak in Fig. 6a. This indicates a rapid decrease in the detected molecular dynamics. The PC → OC phase transition (i.e., on cooling) is characterised by the formation of the hydrogen bond network present in the BCT phase, and thus it is expected that the low IFWS signal below ~ 280 K is due to PC dynamics being frozen by the existence of an extensive hydrogen bond network that immobilises the rotation of the molecules and their hydroxymethyl groups. Figure 7a shows the IFWS data measured at \(\pm\)3 µeV on cooling from 366 K to 100 K for \(Q\) = 0.83 Å−1 at a rate of 1 K min−1, directly comparable to our calorimetry and XRD measurements. Further fits for low- and high-\(Q\) IFWS data can be found in Supplementary Fig. 21. Figure 7b shows the corresponding IFWS data on heating from 240 K to 355 K. We will first discuss the cooling data as it contains a larger temperature range. The peak centred around 200 K in Fig. 7a corresponds to Mode 1, the methyl rotation, as we observed previously in NPG50. Peaks in the IFWS data are attributed to the Lorentzian terms ‘moving through’ the fixed energy window as the associated mode changes in frequency with changing temperature (broadening into the background for heating and narrowing into the elastic line for cooling). The inset plot compares the peak intensity of NPG-PG-PE with that of NPG after normalisation to the low-\(T\) region, showing that the NPG-PG-PE peak is less intense than in NPG. This reduced intensity can be attributed to the solid solution containing 21% fewer methyl groups than NPG, which is consistent with the measured intensity ratio of 0.76 ± 0.02. Fitting the thermal variation of Mode 1 (methyl rotation, blue solid line) as described previously57, we model a temperature dependence by assuming an Arrhenius law for the linewidth (\(\varGamma (Q)\)):
$$\Gamma \left(Q\right)={\Gamma }_{0}\left(Q\right){e}^{-\frac{{E}_{{\rm{a}}}}{{k}_{{\rm{B}}}T}}.$$
(1)
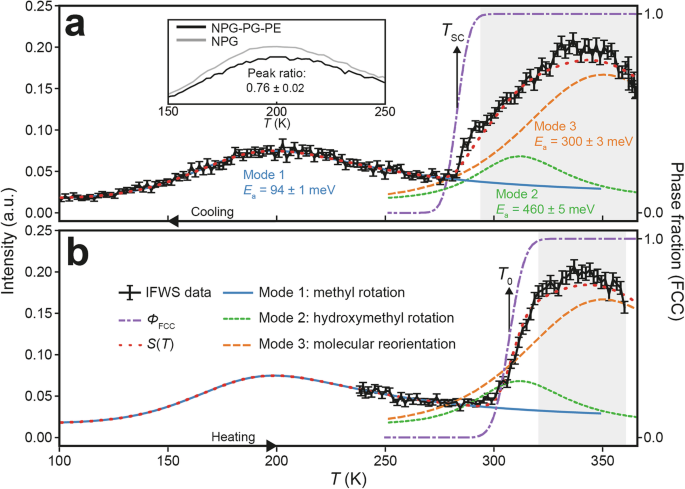
a Inelastic fixed window scan (\(E\) = ±3 µeV, \(Q\) = 0.83 Å−1) analysis on cooling from 366 K to 100 K. All inelastic fixed window scan data corresponds to the left axis. The peak centred around 200 K corresponds to the methyl group rotations (solid blue line). The peak centred around 340 K can be fitted with two modes: hydroxymethyl rotation (solid green line) and molecular reorientation (solid orange line). The purple dashed line corresponds to the fitted FCC phase fraction (\({\varPhi }_{{\rm{FCC}}}\), right axis) obtained from XRD and using this, it is possible to mix the three modes in the phase coexistence region to provide a fit of \(S(T)\) over the entire temperature range (red dotted line). The inset plot compares the methyl rotation peak amplitude between the solid solution and pure NPG. b Inelastic fixed window scan (\(Q\) = 0.83 Å−1) on heating from 240 K to 360 K with the fits obtained from the cooling data in (a). Shaded regions indicate the temperatures at which \({\varPhi }_{{\rm{FCC}}}\) > 0.98, and the material was considered fully in the PC phase.
By restricting fitting of the data to \(T\) < 260 K, we ensure that the phase transition region is not included in the fit, and we calculate an activation energy (\({E}_{a}\)) of 94 ± 1 meV for methyl rotation in the OC phase. The value obtained is close to the activation energy of 100 meV obtained for pure NPG50, demonstrating the consistency of our model. The sharp intensity change observed upon cooling from approximately 300 K to 290 K is attributed to the PC → OC phase transition, during which the rotational modes present in the PC phase become immobile as the extensive hydrogen bond network of the OC phase forms. The peak centred around 340 K contains the intensity contribution from both Mode 2, hydroxymethyl rotation, and Mode 3, molecular reorientation. It is possible to separate the contributions of each mode to the total IFWS intensity in the PC phase. This was achieved by globally fitting all obtained IFWS cooling data with the QENS data at 330 K and 360 K to constrain the intensity ratio of Mode 2 and Mode 3 determined by the QENS fits (see Supplementary Information Note 3 for further details). Furthermore, it is only meaningful to fit the IFWS data in regions where the material is fully in the PC phase. To determine the range of IFWS data to fit we parameterise the FCC phase fraction (\({\varPhi }_{{\rm{FCC}}}\)) by fitting error functions (Supplementary Information Note 2) to the full heating and cooling synchrotron XRD data (Fig. 4).
By fitting the IFWS cooling data for temperatures where \({\varPhi }_{{\rm{FCC}}}\) > 0.98, shown by the grey shaded region in Fig. 7a, we obtain \({E}_{a}\) for Mode 2 (hydroxymethyl rotation, green dashed line) and Mode 3 (molecular reorientation, orange dashed line) of 460 \(\pm\) 5 meV and 300 \(\pm\) 3 meV, respectively. These values are both approximately 50% lower than the corresponding activation energies of the same modes observed experimentally in NPG50. Furthermore, the measured activation energy of hydroxymethyl rotation in NPG-PG-PE is close to that of two hydrogen bonds (~500 meV48), suggesting that the energy barrier to hydroxymethyl rotation is highly correlated with the hydrogen bond network in the ternary solid solution. We note that obtaining precise activation energies of multiple overlapping modes is not trivial and is enabled by our ability to constrain the mode intensity ratios by including the QENS datasets in the global fitting procedure. The reduction in activation energies of Modes 2 and 3 agrees with the observations from the XRD data that the energy landscape of the hydrogen bond network in NPG-PG-PE is significantly altered as compared to NPG. Curiously, the activation energy for the hydroxymethyl rotation is found to be larger than that for the molecular reorientation, as has also been observed experimentally50 and theoretically48 for NPG. Further support for this interpretation is gained from an inspection of the relative magnitudes of the fitted pre-exponential factors (\({\varGamma }_{0}(Q)\)) used in the Arrhenius law in Eq. 1. The full \({\varGamma }_{0}(Q)\) data can be found in Supplementary Fig. 22. Converting the pre-exponential factors to attempt frequencies yields ~140 THz and ~100 GHz for hydroxymethyl rotation and molecular reorientation, respectively. These values are consistent with molecular rotational attempt frequencies around the microwave region58. They demonstrate that although the hydroxymethyl rotation has a higher energy barrier to overcome, it also has an attempt frequency around 3 orders of magnitude greater than that of molecular reorientation, which may reconcile the relative magnitudes of their activation energies. Together, these observations suggest that hydroxymethyl rotation is concurrent with the extinction of the hydrogen bond network and unlocks whole molecular rotation, with its lower activation energy. Therefore, lowering the energy barrier for hydroxymethyl rotation enables access to the large entropy change associated with molecular reorientations at lower temperatures.
Thermodynamically, we do not expect a difference in activation energies or attempt frequencies for cooling versus heating, only a difference in the transition temperature and phase coexistence region. By fixing \({E}_{a}\) and \({\varGamma }_{0}(Q)\) obtained from the fits of the IFWS cooling data, we can assess how well the derived model is able to describe the heating data (Fig. 7b). The purple dashed lines in Fig. 7a, b show the FCC phase fraction (\({\varPhi }_{{\rm{FCC}}}\)) on cooling and heating as determined from the XRD data (Supplementary Information Note 2), respectively, with \({T}_{{\rm{SC}}}\) and \({T}_{0}\) indicated by the vertical black arrows. Here, we present a method to utilise \({\varPhi }_{{\rm{FCC}}}\) to fit the total temperature-dependent quasielastic behaviour (\(S(T)\), given by the red dotted line) through the phase transition for 0.02 <\({\varPhi }_{{\rm{FCC}}}\) < 0.98. To do this, we weighted the previously obtained Lorentzians (\({L}_{1}\), \({L}_{2}\) and \({L}_{3}\)) from fitting the IFWS cooling data by the corresponding phase fraction according to:
$$S\left(T\right)=\left(1-{\varPhi }_{{\rm{FCC}}}\right){L}_{1}+{\varPhi }_{{\rm{FCC}}}\left({L}_{2}+{L}_{3}\right).$$
(2)
As can be seen by the trends in Fig. 7a, b the red dotted lines that describe \(S(T)\) provide good fits for the phase transition regions on both cooling and heating. This highlights the need to consider modes from both the OC and PC phases throughout the broad phase coexistence regions. Note that Mode 1 (methyl rotation) is also active in the PC phase; however, the FWHM linewidth of this mode (~500 μeV50) is well outside that of IN16B’s dynamic range (56 µeV), and as such, we are not directly sensitive to it. The ability to combine XRD, QENS and FWS data in this way provides a powerful tool for describing dynamical mode behaviour even during phase transitions, where phase heterogeneity is significant.
Discussion
The observation that BC properties can be tuned intrinsically is exemplified in Fig. 2. Choosing an NPG:PG ratio of around 60:40 results in a single-phase material that exhibits a \({T}_{0}\) that is 10 K lower than NPG and 50 K lower than PG, around the regime required for BC cooling. The main result of this study is that adding a small amount of a third component, in this case PE, leaves \({T}_{0}\) largely unchanged but drastically improves the reversibility of the BCE as exemplified in Fig. 2d. The improvement is due to an asymmetric increase in the \(\alpha ,\gamma\) phase-coexistence region of the PC → OC phase transition, which results in a significant overlap of latent heat in the hysteresis region, as shown in both Figs. 2c and 6a. Furthermore, the solid solutions of NPG-PG and NPG-PG-PE exhibit enhanced phase coexistence regions, which is more pronounced at higher pressures, as shown in Fig. 2a. This explains the broad temperature range, \(\Delta {T}_{{\rm{span}}}\) ~ 18.0 K, over which NPG-PG-PE exhibits a reversible entropy change. This flexibility provides potential opportunities for multi-component cascading barocaloric devices59, where the composition of the solid solution, and hence the transition temperature, is engineered to vary within a heating/cooling device to improve efficiency.
Our results also reveal the structural changes occurring within the phase coexistence region and the nature of the doping in NPG-PG-PE. The synchrotron XRD data presented in Fig. 5 demonstrates that the OC → PC phase transition exhibits a broadened first-order behaviour within the temperature range 0.96 <\(\frac{T}{{T}_{0}}\) < 0.99, characterised by tracking the anisotropic diffraction broadening parameters \({S}_{400}\) and \({S}_{220}\), indicative of weakened hydrogen bonding in the \({ab}\) plane. This contrasts with the sharply first-order behaviour of isostructural PG, which shows negligible \({S}_{220}\) broadening and a sharp transition of \({S}_{400}\) increasing within a narrow temperature range around \({T}_{0}\) (Supplementary Fig. 15). These observations are also supported by the IFWS data presented in Fig. 7, where we calculate activation energies of the PC modes that are approximately 50% lower than those of NPG. In NPG-PG-PE, the change in quasielastic signal is much more gradual through the phase transition (on both heating and cooling) than pure NPG (Supplementary Fig. 23), due to the significant mixing of OC and PC rotational modes within the phase coexistence region.
The improvement of BC performance in NPG-PG-PE compared to NPG-PG comes primarily from a reduction in thermal hysteresis, which is caused by a shift of the PC → OC phase transition to higher temperatures, as exemplified in the entropy change data in Fig. 2c. The diffraction results (Fig. 5 and Supplementary Fig. 15) indicate that the binary and ternary have increased short-range disorder or strain over a broad temperature range when compared to PG. Furthermore, the ternary exhibits significantly higher strain than the other materials, particularly on cooling (Supplementary Fig. 16). In turn, this suggests that dopant PE molecules, even at 2%, induce further significant local distortions in the host NPG-PG structure. It is tempting to conclude that the local, often transient hotspots observed in IR while cooling the NPG-PG-PE sample correspond to inclusions of higher PE concentration that undergo the PC → OC transition before the surrounding matrix, but are insufficient to trigger a wave of transition throughout the material. A critical question is therefore the location of the PE dopant molecules, which may help explain why such a small quantity of PE has such a profound effect on the phase-change behaviour. It is conceivable that the PE molecules could be incorporated into the BCT structure interstitially. However, our XRD data support direct substitution of NPG molecules for PE. In accordance with our initial hypothesis, direct substitution is facilitated by the structural similarities between the neopentane molecules.
The substitutional model is further evidenced by our single crystal electron diffraction measurements (Supplementary Information Note 1 and Supplementary Figs. 17 and 18), where fractional occupation of hydroxymethyl and methyl groups in the refined structures is in line with the composition of NPG-PG-PE, assuming a substitutional inclusion of NPG and PE (as shown in Supplementary Tables S1–S6). On a substitutional site, PE is directly involved in the hydrogen bond network and so would be expected to affect bonding within the \({ab}\) plane, and hence explain the enhanced broadening of \({hk}0\) type reflections in NPG-PG-PE compared to NPG-PG and PG. It may simply be localised strain around the substitutional PE molecules that facilitates localised phase changes and thereby extends the phase coexistence region, specifically on cooling.
Our strategy has been to employ substitutional organic doping of molecular crystals with similar molecules as a general method of improving their thermal and pressure reversibility. Looking forward, this strategy will also be applicable to, but is not limited to, the adamantane PC family, where functional substitution of H atoms with more electronegative groups such as -Cl and -Br results in materials that exhibit a colossal BCE at distinct temperatures12,14,46. These functional substitutions do not significantly change the structure of the PC phase but may drastically alter the orientational molecular dynamic processes41,43, OC structure60 and barocaloric performance46. As such, the adamantane PC family may be suitable for BC enhancement through small compositional doping of organic derivatives.
Conclusions
We have demonstrated a strategy to improve plastic crystal barocaloric performance with respect to operating temperature (\({T}_{0}\)), reversible temperature span (\(\varDelta {T}_{{\rm{span}}}\)) and reversible entropy change (\(|\varDelta {S}_{{\rm{it}}}|\)). Using NPG as an exemplar, and by forming the solid solution of NPG-PG-PE in a molecular ratio of 60:38:2, we reduce \({T}_{0}\) by 10 K and increase \({{\rm{RC}}}_{{\rm{rev}}}\) by a factor of seventy, arising from the increase in \(\varDelta {T}_{{\rm{span}}}\) by a factor of twenty and increase \(|\varDelta {S}_{{\rm{it}}}|\) by a factor of seven at 1 kbar. This improvement comes from an enduring OC-PC phase coexistence that broadens the phase transition between low- and high-entropy phases at both ambient and elevated pressures. We present structural and dynamical evidence for a broad phase coexistence region that is characterised by a disrupted hydrogen bond network. We propose that disrupting the network through substitutional doping is a strategy for improving the reversibility of the barocaloric phase transition. Our study showcases a new strategy for designing practicable molecular crystal barocalorics with tuneable operating temperatures and reduced hysteresis. Given the vast compositional phase space for molecular crystal solid solutions, the prospects for applying this strategy to other molecular systems are exciting.
Methods
Sample preparation
Powder samples of NPG, PG and PE (99% purity, Sigma-Aldrich) were used as received. Powders were mixed and then dissolved in ethanol before being left to recrystallize under ambient conditions for a period of 1 week. Each sample was then subjected to thermal treatment consisting of heating to 350 K for 30 min and cooling to 265 K for 24 h, which was to ensure that the samples were in their low temperature phase. Samples were then stored in sealed containers at room temperature. Some of the NPG-PG-PE sample was stored in the freezer at 265 K for 3 months, and its T0 value, obtained from ambient pressure calorimetry, was compared to the sample stored at room temperature to confirm that no phase segregation had occurred.
Calorimetry
All calorimetry measurements and analysis reported here have been performed on samples that have been thermally cycled around the OC → PC transition at least 10 times prior to remove virgin effect variations28. Supplementary Fig. 2c demonstrates the stability of the calorimetric behaviour of NPG-PG-PE after the first thermal cycle. Calorimetric measurements under ambient pressure were obtained using a TA Instruments DSC 250 at a temperature-ramping rate of \(\frac{{|dT|}}{{dt}}\) = 0.5 K min−1 with sample masses between 5 and 15 mg. Calorimetric measurements under applied pressure used sample masses of between 45 and 70 mg and were performed using a Setaram μDSC7 Evo with an applicable hydrostatic pressure limit of up to 1200 bar, using high-purity (99.9999%) nitrogen gas as the pressure-transmitting medium. A temperature-ramping rate of \(\frac{{|dT|}}{{dt}}\) = 1.2 K min−1 was used to obtain isobaric calorimetric signals, \({(\frac{{dq}}{\left|{dT}\right|})}_{p}\), within a temperature range 260 K <\(T\) < 340 K, which were then background-subtracted. The integral areas of the residual transition peaks were then obtained to evaluate the barocaloric effect in the samples quasi-directly using
$$\left|\Delta {S}_{{\rm{it}}}\right|\left(T,{p}_{{\rm{i}}}\to {p}_{{\rm{f}}}\right)={\int }_{{T}_{1}}^{T}\left[{\left(\frac{{dq}\left({T}^{{\prime} },{p}_{{\rm{f}}}\right)}{\left|{\rm{d}}{T}^{{\prime} }\right|}\right)}_{p}-{\left(\frac{{dq}\left({T}^{{\prime} },{p}_{{\rm{i}}}\right)}{\left|{\rm{d}}{T}^{{\prime} }\right|}\right)}_{p}\right]\frac{{\rm{d}}{T}^{{\prime} }}{{T}^{{\prime} }}$$
(3)
where \(|\Delta {S}_{{\rm{it}}}|(T,\,{p}_{i}\to {p}_{f})\) is the isothermal entropy change driven by changes in pressure from \({p}_{i}\) (initial pressure) to \({p}_{f}\) (final pressure) at temperature \(T\). \({T}_{1}\) is an arbitrary starting temperature selected below the phase transition at zero applied pressure and \(T\mbox{'}\) is the dummy variable of integration. We use lowercase \(q\) for heat to avoid confusion with the scattering variable \(Q\) in the neutron spectroscopy data.
Infra-red imaging
Samples were prepared for imaging by pelletising ~ 0.1 g of material into a pellet with a 13 mm diameter to create a disk of sample <1 mm in thickness. This was placed on a glass slide and heated to 330 K for 5 minutes using a Linkam PE120 Peltier heating and cooling stage. The sample was then allowed to cool at a rate of 5 K min−1 and imaged using an Optris PI640 IR microscope camera as it went through its phase transition on cooling. Images were acquired every 2 s, and difference images were constructed by subtracting subsequent frames from preceding ones.
Synchrotron diffraction
Synchrotron powder diffraction was performed on the I11 beamline at Diamond Light Source, UK, using \({E}_{{\rm{i}}}\) = 15 keV. Samples were packed in 0.5 mm diameter borosilicate capillaries. Temperature was controlled using the Cryostream, and data were collected continuously with a ramp rate of 1 K min−1 with the position sensitive detectors (PSD). All samples were thermally cycled four times in-situ before measurement, to account for virgin effects. Rietveld refinement was performed using GSAS-II61. The BCT phase of both PG and NPG-PG-PE was modelled using the published62 crystal structure of isostructural PE as a starting point. Anisotropic strain parameters, \({S}_{{hkl}}\), were determined within the GSAS-II software. Temperature-dependent diffraction peak broadening parameters, \({S}_{{hkl}}\), for \({hkl}\) reflections were fitted for each sample according to \({\Gamma }_{{\rm{hkl}}} \sim {S}_{{\rm{HKL}}}{h}^{H}{k}^{K}{l}^{L}\), where \({\varGamma }_{{hkl}}\) is the diffraction peak full width at half maximum43. \({S}_{400}\), \({S}_{004}\) and \({S}_{220}\) parameters were required for NPG-PG-PE refinement, whereas, only \({S}_{400}\) and \({S}_{004}\) parameters gave positive, non-negligible and stable values during PG refinement. Supplementary Fig. 24 shows a comparison of using anisotropic verses isotropic strain models. For the FCC phase, true Rietveld refinement was not attempted due to the significant disorder of the molecule. In order to extract the relative fractions of the BCT and FCC phases, the scattering power of the atoms in the FCC unit cell was modelled using a dummy carbon atom with the position and thermal parameter allowed to refine, modelling the two peaks visible in the diffractogram. The resulting refined phase fraction of the FCC phase as a function of temperature on heating and cooling was used in the QENS analysis. Transition temperatures were defined as the temperatures at which the sample had transitioned halfway, given by the phase fraction fitted from XRD refinements.
Quasielastic neutron scattering
Neutron scattering experiments were performed on the IN16B spectrometer at the Institut Laue–Langevin (ILL), France. IN16B is a backscattering spectrometer and was used in a standard configuration with strained Si111 Doppler monochromator and analysers, yielding a FWHM energy resolution of 0.75 μeV, dynamic range of \(\pm\) 0.028 meV and \(Q\)-range of 0.19 to 1.89 Å−1. QENS measurements were obtained using scan times of 2 h. All QENS measurements were obtained on heating. FWS measurements were performed during a temperature ramp of approximately 0.5 K min−1 with alternating acquisitions of elastic (30 s) and inelastic intensity at 3 µeV energy transfer (90 s). The sample was thermally cycled five times immediately before being loaded into a measurement can to account for virgin effects. Approximately 0.5 g of sample was loaded into an aluminium can with annular geometry for measurements on IN16B. Empty can and vanadium standard measurements were acquired using the same sample geometry and used to correct the sample data. Spectrometer resolution measurements were obtained at 2 K.
All data were analysed using Mantid v6.6.0 software63. Full details on QENS/FWS analysis and fitting can be found in Supplementary Information Note 3.
Data availability
The high-pressure calorimetry, X-ray diffraction and infra-red imaging data that support the findings of this study are available at https://doi.org/10.5525/gla.researchdata.2130. The quasielastic neutron scattering data is available at https://doi.org/10.5291/ILL-DATA.7-02-227.
References
Kitanovski, A., Plaznik, U., Tomc, U. & Poredoš, A. Present and future caloric refrigeration and heat-pump technologies. Int. J. Refrig. 57, 288–298 (2015).
Moya, X. & Mathur, N. D. Caloric materials for cooling and heating. Science 370, 2020 (1979).
Boldrin, D. Fantastic barocalorics and where to find them. Appl. Phys. Lett. 118, 170502 (2021).
Cirillo, L., Greco, A. & Masselli, C. Cooling through barocaloric effect: a review of the state of the art up to 2022. Thermal Sci. Eng. Progress 33, 101380 (2022).
Schipper, J. et al. On the efficiency of caloric materials in direct comparison with exergetic grades of compressors. JPhys Energy 5, 045002 (2023).
Cirillo, L., Greco, A. & Masselli, C. The application of barocaloric solid-state cooling in the cold food chain for carbon footprint reduction. Energies 16, 6436 (2023).
Sun, Y. et al. Materials with the barocaloric effect for solid-state refrigeration. J. Mater. Chem. A 13, 6152–6175 (2025).
Lloveras, P. & Tamarit, J. L. Advances and obstacles in pressure-driven solid-state cooling: a review of barocaloric materials. MRS Energy Sustain. 8, 3–15 (2021).
Lloveras, P. et al. Colossal barocaloric effects near room temperature in plastic crystals of neopentylglycol. Nat. Commun. 10, 1–7 (2019).
Li, B. et al. Colossal barocaloric effects in plastic crystals. Nature 567, 506–510 (2019).
Aznar, A. et al. Reversible and irreversible colossal barocaloric effects in plastic crystals. J. Mater. Chem. A Mater. 8, 639–647 (2020).
Aznar, A. et al. Reversible colossal barocaloric effects near room temperature in 1-X-adamantane (X=Cl, Br) plastic crystals. Appl. Mater. Today 23, 101023 (2021).
Zhang, K. et al. Colossal barocaloric effect in carboranes as a performance tradeoff. Adv. Funct. Mater. 32, 2112622 (2022).
Salvatori, A. et al. Colossal barocaloric effects in adamantane derivatives for thermal management. APL Mater. 10, 111117 (2022).
Salvatori, A. et al. Large barocaloric effects in two novel ferroelectric molecular plastic crystals. J. Mater. Chem. A Mater. 11, 12140–12150 (2023).
Zeng, M. et al. Colossal reversible barocaloric effects in a plastic crystal mediated by lattice vibrations and ion diffusion. Adv. Sci. 11, 2306488 (2024).
Piper, S. L. et al. Organic ionic plastic crystals having colossal barocaloric effects for sustainable refrigeration. Science 387, 2025 (1979).
Imamura, W. et al. Supergiant barocaloric effects in acetoxy silicone rubber over a wide temperature range: great potential for solid-state cooling. Chin. J. Polym. Sci. 38, 999–1005 (2020).
Lünser, K. et al. Elastocaloric, barocaloric and magnetocaloric effects in spin crossover polymer composite films. Nat. Commun. 15, 1–10 (2024).
Li, J. et al. Colossal reversible barocaloric effects in layered hybrid perovskite (C10H21NH3)2MnCl4 under low pressure near room temperature. Adv. Funct. Mater. 31, 2105154 (2021).
Seo, J. et al. Colossal barocaloric effects with ultralow hysteresis in two-dimensional metal–halide perovskites. Nat. Commun. 13, 2536 (2022).
García-Ben, J. et al. Unveiling barocaloric potential in organometallic-sandwich compounds [Cp2M][PF6] (M: Fe3+, Co3+). J. Mater. Chem. A Mater. 12, 23751–23760 (2024).
Seo, J. et al. Barocaloric effects in dialkylammonium halide salts. J. Am. Chem. Soc. 146, 2736–2747 (2024).
Abas, N. et al. Natural and synthetic refrigerants, global warming: a review. Renew. Sustain. Energy Rev. 90, 557–569 (2018).
Takeuchi, I. & Sandeman, K. Solid-state cooling with caloric materials. Phys. Today 68, 48–54 (2015).
Lloveras, P. Barocaloric Effects in the Solid State https://doi.org/10.1088/978-0-7503-4690-0 (IOP Publishing, 2023).
Lilley, D., Lau, J., Dames, C., Kaur, S. & Prasher, R. Impact of size and thermal gradient on supercooling of phase change materials for thermal energy storage. Appl. Energy 290, 116635 (2021).
Rendell-Bhatti, F., Boldrin, D., Dilshad, M., Moya, X. & MacLaren, D. A. Understanding variations of thermal hysteresis in barocaloric plastic crystal neopentyl glycol using correlative microscopy and calorimetry. J. Phys. Energy 6, 025020 (2024).
Font, J., Muntasell, J., Navarro, J., Tamarit, J. L. & Lloveras, J. Calorimetric study of the mixtures PE/NPG and PG/NPG. Sol. Energy Mater. 15, 299–310 (1987).
Salud, J., López, D. O., Barrio, M. & Tamarit, J. L. Two-component systems of isomorphous orientationally disordered crystals. Part 1: packing of the mixed crystals. J. Mater. Chem. 9, 909–916 (1999).
Wang, X., Lu, E., Lin, W. & Wang, C. Micromechanism of heat storage in a binary system of two kinds of polyalcohols as a solid-solid phase change material. Energy Convers. Manag. 41, 135–144 (2000).
Mishra, A., Talekar, A., Chandra, D. & Chien, W. M. Ternary phase diagram calculations of pentaerythritol-pentaglycerine- neopentylglycol system. Thermochim. Acta 535, 17–26 (2012).
Singh, H. et al. Continuous solid-state phase transitions in energy storage materials with orientational disorder - Computational and experimental approach. Energy 91, 334–349 (2015).
Serrano, A., Duran, M., Dauvergne, J. L., Doppiu, S. & Del Barrio, E. P. Tailored transition temperature plastic crystals with enhanced thermal energy storage capacity. Sol. Energy Mater. Sol. Cells 220, 110848 (2021).
Li, F., Niu, C., Xu, X., Li, M. & Wang, H. The effect of defect and substitution on barocaloric performance of neopentylglycol plastic crystals. Appl. Phys. Lett. 121, 223902 (2022).
Liu, Y. et al. Giant barocaloric effect in neopentylglycol-graphene nanosheets composites with large thermal conductivity. Mater. Res. Lett. 10, 675–681 (2022).
Li, F. et al. Predicting large comprehensive refrigeration performance of plastic crystals by compositing carbon architectures for room temperature application. Appl. Phys. Lett. 123, 183902 (2023).
Dai, Z. et al. Plastic crystal neopentyl glycol/multiwall carbon nanotubes composites for highly efficient barocaloric refrigeration system. J. Therm. Sci. 33, 383–393 (2024).
Chandra, D., Barrett, C. S. & Benson, D. K. X-ray diffraction studies of solid solutions of pentaglycerine-neopentylglycol. Adv. X-ray Anal. 32, 609–616 (1988).
Negrier, P., Barrio, M., Tamarit, J. L. & Mondieig, D. Polymorphism in 2-X-adamantane derivatives (X = Cl, Br). J. Phys. Chem. B 118, 9595–9603 (2014).
Negrier, P. et al. Polymorphism of 1,3-X-adamantanes (X = Br, OH, CH3) and the crystal plastic phase formation ability. CrystEngComm 22, 1230–1238 (2020).
Santiago-Sosa, O. et al. Phase change enthalpies of some monosubstituted derivatives of adamantane: an experimental and theoretical study. Struct. Chem. 1, 3 (2022).
Meijer, B. E. et al. Dynamics in the ordered and disordered phases of barocaloric adamantane. Phys. Chem. Chem. Phys. 25, 9282 (2023).
Moya, X., Kar-Narayan, S. & Mathur, N. D. Caloric materials near ferroic phase transitions. Nat. Mater. 13, 439–450 (2014).
Somodi, C. B., McCormick, K., Tabor, D. P., Pentzer, E. & Shamberger, P. J. Kinetics of the plastic crystal transition in neopentyl glycol. J. Appl. Phys. 135, 145101 (2024).
Santos, A. P. et al. Effect of functional groups on neopentane and adamantane derivative plastic crystal phase transition in molecular simulations. J. Phys. Chem. B 129, 9745–9755 (2025).
Stephens, P. W. Phenomenological model of anisotropic peak broadening in powder diffraction. J. Appl. Crystallogr. 32, 281–289 (1999).
Li, F. B. et al. Understanding colossal barocaloric effects in plastic crystals. Nat. Commun. 11, 1–8 (2020).
Meijer, B. E., Cai, G., Demmel, F., Walker, H. C. & Phillips, A. E. Pressure dependence of rotational dynamics in barocaloric ammonium sulfate. Phys. Rev. B 106, 064302 (2022).
Rendell-Bhatti, F. et al. Direct observation of thermal hysteresis in the molecular dynamics of barocaloric neopentyl glycol. ACS Appl. Energy Mater.8, 4793-4802 (2025).
Kruteva, M. Dynamics studied by Quasielastic Neutron Scattering (QENS). Adsorption 27, 875–889 (2021).
Line, C. M. B., Winkler, B. & Dove, M. T. Quasielastic incoherent neutron scattering study of the rotational dynamics of the water molecules in analcime. Phys. Chem. Min. 21, 451–459 (1994).
Silvi, L., Röhm, E., Fichtner, M., Petry, W. & Lohstroh, W. Hydrogen dynamics in β-Mg(BH4)2 on the picosecond timescale. Phys. Chem. Chem. Phys. 18, 14323–14332 (2016).
Songvilay, M. et al. Decoupled molecular and inorganic framework dynamics in CH3NH3PbCl3. Phys. Rev. Mater. 3, 125406 (2019).
Seven, O. & Bolte, M. CCDC 832035: experimental crystal structure determination. https://doi.org/10.5517/ccwxsvd (2011).
Sanuy, A. et al. Molecular origins of colossal barocaloric effects in plastic crystals. https://doi.org/10.48550/arXiv.2501.14403 (2025).
Frick, B., Combet, J. & Van Eijck, L. New possibilities with inelastic fixed window scans and linear motor Doppler drives on high resolution neutron backscattering spectrometers. Nucl. Instrum. Methods Phys. Res. A 669, 7–13 (2012).
Colombo, L. Molecular vibrations and rotations. in Atomic and Molecular Physics 2 edn 7.1-7.16 https://doi.org/10.1088/978-0-7503-5734-0ch7 (IOP Publishing, 2023).
Torelló, À, Tamarit, J. L. & Lloveras, P. Finite-element study of a high-performance cascaded barocaloric cooler. Appl. Therm. Eng. 280, 128055 (2025).
Salzillo, T., Girlando, A. & Brillante, A. Revisiting the disorder-order transition in 1-X-adamantane plastic crystals: Rayleigh wing, boson peak, and lattice phonons. J. Phys. Chem. C. 125, 7384–7391 (2021).
Toby, B. H. & Von Dreele, R. B. GSAS-II: the genesis of a modern open-source all purpose crystallography software package. J. Appl. Crystallogr. 46, 544–549 (2013).
Zhou, Q. et al. A clustering-triggered emission strategy for tunable multicolor persistent phosphorescence. Chem. Sci. 11, 2926–2933 (2020).
Arnold, O. et al. Mantid—Data analysis and visualization package for neutron scattering and μ SR experiments. Nucl. Instrum. Methods Phys. Res. A 764, 156–166 (2014).
Momma, K. & Izumi, F. VESTA 3for three-dimensional visualization of crystal, volumetric and morphology data. J. Appl. Cryst. 44, 1272–1276 (2011).
Acknowledgements
We acknowledge beam time awarded by the ILL neutron source (proposal 7-02-227). We acknowledge Diamond Light Source for time on I11 under proposal CY40567. For the purpose of open access, the authors have applied a Creative Commons Attribution (CC BY) licence to any Author Accepted Manuscript version arising from this submission. This work was financially supported by an EPSRC grant (EP/V042262/1), a Carnegie Research Incentive Grant (RIG013328) and SGR-00343 Project (Catalonia), by grant PID2023-146623NB-I00 funded by MICIU and by ERDF/EU and is part of Maria de Maeztu Units of Excellence Programme CEX2023-001300-M funded by MCIN/AEI (10.13039/501100011033). We acknowledge an EPSRC strategic equipment funding grant (EP/X030083/1).
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Communications Materials thanks the anonymous reviewers for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Rendell-Bhatti, F., Dilshad, M., Beck, C. et al. Enhanced reversible barocaloric effect at low pressure in neopentyl plastic crystal solid solutions. Commun Mater 7, 72 (2026). https://doi.org/10.1038/s43246-026-01084-2
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s43246-026-01084-2