Introduction
Cryopreservation by vitrification, or the stabilization of biological matter in the glassy state at low-temperature, promises to realize broad transformations of the global bio-cold chain, enabling the banking of transplantable organs and tissues1,2, conservation of threatened flora and fauna3,4, stabilization of volatile pharmaceuticals5,6, etc. However, while the first successful vitrification and transplantation of a mammalian organ was recently achieved at the rodent scale2, significant physical challenges remain in the translation of vitrification approaches to increasingly large-volume biological systems.
These challenges have varied origins in the complex multiphysics of vitrification— loading of chemically diverse vitrification solutions into biological tissues presents a unique and complicated mass transfer problem; reducing the critical cooling and warming rates required to avoid deleterious ice formation en route to the desired storage temperature requires manipulation of nucleation kinetics and growth phenomena; and the achievability of these cooling and warming rates is limited by the thermal transport properties of the sample7,8. One physical challenge that is both unique to vitrification (as opposed to slow-freezing methods) and increasingly prominent at the volume scales relevant to human organs and tissues is the management of thermal stresses, and the catastrophic mechanical failure modes (i.e. cracking) that can accompany them9,10,11,12,13.
Thermal stress during cryopreservation by vitrification is driven by the unfavorable pairing of the high critical cooling/warming rates often needed to achieve aqueous glass formation and the poor thermal diffusivity of biological systems of interest, which can lead to significant thermal gradients across a sample during temperature cycling. These gradients cause differing portions of the sample to expand or contract at differing rates, thereby generating stresses that may quickly approach the low failure strength of brittle aqueous glasses14.
Significant recent work has investigated transport-based methods by which to decrease the risk of thermal stress cracking during vitrification of whole organs and large tissues. Drawing from classical concepts in metallurgy, recent advances have yielded thermal processing approaches (annealing, rate-optimization) and geometric strategies to manage thermal stresses during cooling11,12,13. Likewise for rewarming, many recent works have proposed photothermal and electromagnetic strategies to enable higher warming rates and greater temperature uniformity, seeking to replace conventional convective warming2,15. However, given the overwhelming dominance of a narrow range of established cryoprotectant solutions over the field of organ vitrification16, these works have investigated transport-based thermal stress management within only a small palette of chemistries, and the potential role of solution chemical thermodynamics itself remains understudied.
Cryomacroscope image acquisition platform and deep learning image segmentation architecture. (A) A custom vitrification cassette containing a 40 µl aqueous sample is plunged into liquid nitrogen, held for three minutes, then promptly mounted on the optical tower shown at left and imaged. Schematic drawn in Adobe Illustrator 2025 (www.adobe.com/products/illustrator). (B) These images are then processed using trained semantic segmentation deep learning algorithms in order to segment the image area into the cavity, crack, glass, and background domains.
In particular, the vast majority of published works have studied vitrification chemistries yielding an extremely narrow band of glass transition temperatures \(\:{T}_{g}\) (approximately − 120 °C to -130°)7,16. While many thermophysical aspects of solutions with glass transitions in this range have been studied as they pertain to thermal stress cracking, the role of the glass transition temperature itself, to our knowledge, has not. In light of intriguing recent discoveries of the fundamental entanglement between glass transition temperature and thermal expansion17, we hypothesize that the glass transition temperature itself may prove a dominant factor in the development of thermal stresses during vitrification of aqueous systems.
In this work, we integrate a custom cryomacroscope experimental platform with image segmentation-based machine learning analyses and thermomechanical finite element simulations to explore the effect of glass transition temperature on stress cracking during vitrification of four binary solutions relevant to cryopreservation. We demonstrate experimentally and computationally that, through a suite of multiphysics effects anchored upon the inverse relationship between \(\:{T}_{g}\) and thermal expansion coefficient (α) demonstrated by Lunkenheimer et al.17, solutions with higher glass transition temperatures experience lower stress and reduced cracking compared to those with lower \(\:{T}_{g}\), when thermally cycled to and from liquid nitrogen temperatures under identical conditions. These insights suggest that the commercial vitrification solutions dominating the field for the past several decades may be uniquely ill-suited to the avoidance of thermal stress, and that manipulation of solution glass transition temperature may open new routes by which to relieve the practical constraints imposed by thermal stresses on organ cryopreservation.
Results
Theoretical considerations
Thermal stress, though in practice a function of several interacting thermophysical phenomena (e.g. thermal diffusivity, temperature-dependent viscosity), exists due only to one: thermal expansion. In the limit of zero thermal expansion/contraction, an unconstrained material will experience zero thermal stress, regardless of its other thermophysical properties, the rate of temperature change, etc. Thus, as a naïve starting point from which to examine the characteristics of a solution that may reduce the proclivity of that solution for cracking during vitrification, we may seek characteristics that affect thermal expansion.
Lunkenheimer and colleagues recently demonstrated that the glass transition temperature itself is one such characteristic. Mirroring the famous Lindemann criterion for crystalline materials, which relates the equilibrium melting point of those materials to their thermal expansion by positing a fundamental limit on the amount to which crystalline bonds may stretch before breaking, Lunkenheimer et al.17 demonstrated across disparate classes of materials that the thermal expansion coefficient and glass transition temperature are also inversely related.
Example original and processed images of vitrified solution samples. (a) 49% DMSO; \(\:{T}_{g}\) = -131 °C. (b) 79% glycerol; \(\:{T}_{g}\) = -102 °C. (c) 65% xylitol; \(\:{T}_{g}\:\)= -87 °C. (d) 63% Sucrose; \(\:{T}_{g}\) = -82 °C. Processed images (bottom row) are segmented via deep learning into crack (red), glass (i.e. non-cracked sample area, yellow), cavity (white), and background (green) regions.
Throughout this work, we will analyze aqueous glass cracking through the lens of the underlying thermal stress required, and its relationship to solution properties. However, we note that this is but one approach, and one that prioritizes the conditions needed for initial fracture (e.g. stress and strain) rather than the properties dictating how that fracture will proceed (e.g. fracture toughness). This choice is motivated by our ultimate interest in cryopreservation, wherein cracking of any extent is unacceptable, and thus modulation of the thermal stress profile itself is of paramount interest.
Experimental platform and protocol
In order to generate quantitative insights into thermal stress cracking, we developed a simple cryomacroscope platform, shown in Fig. 1.A. In brief, this device is comprised of a vitrification cassette, an observation tower, and a camera. The vitrification cassette (Fig. 1.A, labeled) is comprised of a 40 µl aqueous sample held inside an anodized Al-7075 washer (inner diameter = 13 mm, thickness = 1.5 mm) and clamped between optically transparent acrylic disks within a 3D-printed PLA frame. This aqueous sample volume ensures the presence of an initial air cavity within the cassette and an according freely-deforming liquid-gas interface, which is essential for reproducing the boundary conditions relevant to bulk-scale organ and tissue vitrification18,19.
Four binary solutions of water and organic molecules of relevance to cryobiology with glass transition temperatures spanning > 50 °C were selected for study: 49 wt% DMSO (\(\:{T}_{g}\) = -131 °C); 79 wt% Glycerol (\(\:{T}_{g}\) = -102 °C), 65 wt% Xylitol (\(\:{T}_{g}\) = -87 °C), and 63 wt% Sucrose (\(\:{T}_{g}\) of -82 °C). These concentrations were chosen to ensure complete sample vitrification and eliminate the risk of partial crystallization during cooling, allowing us to isolate cracking behavior arising solely from stresses within the homogeneous glassy state. Glass transition temperatures for each solution were measured optically to good agreement with the literature20,21,22,23, as described in previous work24 and detailed in SI Note 4.
To vitrify the sample, the entire cassette is plunge-submerged into liquid nitrogen in a vertical orientation and held for 3 min. The cassette is then removed from nitrogen and placed on an optical tower at room temperature (Fig. 1.A), positioned between a 144-LED ring light source and a 4 K-resolution camera (ELP-USB4KHDR01) outfitted with a 100X magnification lens (Waveshare Electronics). A variable neutral density filter (K&F Nano X) is mounted between the cassette and the camera. At approximately one minute after removal from nitrogen (i.e. one minute of warming via natural convection at room temperature), 20 images of the glass are captured, as shown in Fig. 2. For each solution, n = 18–20 independent trials were performed (additional protocol details in the Methods section). In each of the > 80 vitrification-rewarming experiments performed, care was taken to plunge and mount the cassette in a consistent fashion, to ensure similar cooling and warming rates between trials.
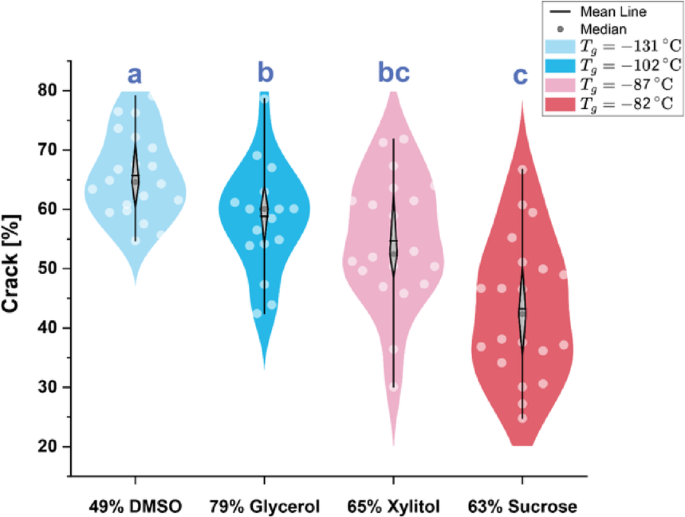
Percentage of sample area identified as cracks via deep learning analysis. Violin plots represent outcomes for n = 20 independent trials (i.e. separate aqueous samples and vitrification cycles) per solution, except for 79% glycerol, which includes n = 18. The median is marked by the central gray circle, the mean is marked by the horizontal gray line, and the vertical lines within each plot indicate the 25th and 75th percentiles.
The acquired images were processed using trained semantic segmentation deep learning algorithms to quantify the extent of cracking area, evaluated on an areal basis, and the geometric evolution of the initial air cavity. In our training model, DeepLab v3 + network was configured with ResNet-50 as the encoder, with the ASPP module used to capture features at multiple scales25,26. The architecture of the deep learning process is shown in Fig. 1.B. The final validation results showed a loss of 0.0424 and an accuracy of 98.32%. Details of the deep learning model and training process are relayed in Supplementary Notes 5.
We note that this mode of crack quantification is imperfect, given the 3D dimensional nature of potential crack phenomena and the 2D nature of the analyzed images. However, because we have chosen a cassette geometry that approaches 2D idealization (sample diameter ~ 10x sample thickness), and because the analysis is consistent across samples, we argue that this approach is sufficient to capture relative differences between samples. We note further that because visual access to the sample is restricted during the nitrogen plunge, we are unable to discern whether the observed cracking occurs during cooling or immediately upon the initiation of rewarming (though we consider the latter more likely). However, the precise moment at which it occurs does not affect our downstream analysis or conclusions, which focus on the accumulation of the thermal stresses that precede crack initiation.
Experimental evidence that higher glass transition temperatures produce less cracking
Figure 2 features representative original and analyzed images for each solution, wherein the image is segmented into one of four categories (cracked, cavity area, uncracked glass, and background areas) and the area occupied by each category is reported as a percentage of the total image area. In order to quantify the degree of cracking observed in the sample, we then normalized the cracked area by the total glass area (i.e. the sum of the areas of the cracked and uncracked glass categories, without the cavity or background contributions). Details and examples of original and analyzed images are presented in Supplementary Video 1.
In Fig. 3, we show extracted crack areas, grouped by solution, aggregated for n = 20 samples per solution and visualized as violin plots with embedded box plots. These box plots highlight the median, mean, and the 25th and 75th percentiles. Pairwise ANOVA tests were calculated to probe statistical significance, with the marked letters indicating statistically similar group means (p > 0.05). Mean and median values within each group were found to be very close, indicating relatively symmetrical distributions, and no strong multi-modalities in the distributions were observed.
As predicted, the mean crack area was found to decrease with increasing glass transition temperature in a statistically significant manner. Intriguingly, the range of observed crack areas was also found to increase with Tg, which appears consistent with the interpretation of crack formation as a stochastic activated process, which (like nucleation and other activated processes) should grow more deterministic with increasing driving force (here thermal stress)27,28. These observations appear consistent with the findings of Lunkenheimer et al.17 suggesting that thermal expansion decreases with increasing glass transition temperature, and are furthermore consistent with previous observations that aqueous solutions of small molecular solvent cryoprotectants such as ethylene glycol (which have glass transition temperatures nearing that of water, typically <-120 °C) crack much more readily than those of larger sugar-based molecules or polymers (which possess higher glass transition temperatures2930). However, the suite of multiphysics phenomena driving thermomechanical stress in vitrifying systems of course transcends simple thermal expansion14. As such, we proceeded to computationally interrogate the plausible dependences of other relevant physical effects on glass transition temperature.
Computational interrogation of T g-dependent thermomechanics in vitrifying systems
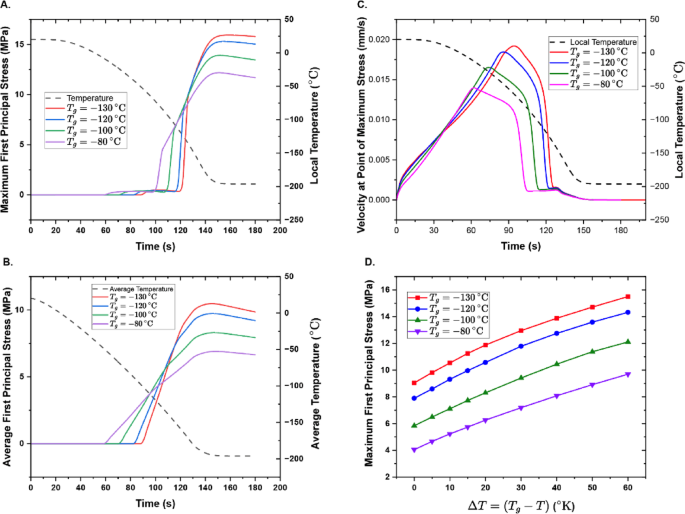
Thermomechanics analyses of first principal stresses in a model vitrification solution with various assumed glass transition temperatures. (a) Maximum first principal stress during cooling, sampled at the bottommost point of the liquid-cavity interface, where deformation is greatest. (b) Average first principal stress throughout the entire sample during cooling. (c) Velocity at the point of maximum stress (bottommost point of the liquid-cavity interface) during cooling. (d) Maximum principal stress plotted as a function of difference in temperature (ΔT) from the glass transition temperature \(\:{T}_{g}\).
Following the approach developed by Rabin18, we conducted a MATLAB-COMSOL thermomechanical finite element analysis of the glass formation process within the cryomacroscope sample geometry shown in Fig. 1.A. This analysis couples a conductive and natural convective heat transport model, a Navier-Stokes creeping flow fluid mechanics model, and a Maxwell fluid model with a single viscoelastic branch in order to capture the evolution of the stress-strain field in the sample during vitrification. Consistent with previous work, and given the much lesser tensile strengths of brittle aqueous glasses as compared to their compressive strengths, the first principal stress is studied as the plausible predictor of glass fracture. To overcome the significant dearth of cryogenic-temperature thermophysical property data available for most of the solutions studied here, we conduct this analysis on a model glass-forming system that employs the properties (temperature-dependent heat capacity, temperature-dependent thermal conductivity, room-temperature reference density, room-temperature reference viscosity, room-temperature reference thermal expansion coefficient) of 7.05 M DMSO31 as a reference basis, then parameterizes the thermal expansion coefficient and temperature-dependent viscosity as functions of glass transition temperature using the relations of Lunkenheimer et al.17 (thermal expansion) and Trejo Gonzalez et al.32 (viscosity). The computational approach is described in detail in Supplementary Information Note 1, and a flow diagram of the logical pipeline is shown in Figure S2.
In brief, Lunkenheimer et al.17 identified that the relation \(\:{\alpha\:}_{l}{T}_{g}\approx const.\) holds across many classes of materials, including aqueous and organic glasses, implying that the thermal contraction of the liquid \(\:{\alpha\:}_{l}\) will decrease with increasing glass transition temperature \(\:{T}_{g}\). Furthermore, they demonstrated that the thermal contraction of the liquid and glassy phases maintain a universal ratio \(\:\frac{{\alpha\:}_{l}}{{\alpha\:}_{g}}\approx\:3\) across materials. Trejo Gonzalez et al.32 have proposed a relation for viscosity also normalized to the glass transition temperature, \(\mu = \gamma \exp\left[ \left(28.75 - \ln \gamma \right) \left( \frac{T_g}{T} \right)^{\beta} \right]\), where γ and β are fitting constants and \(\:{T}_{g}\) is the only additional material input. With these two relations, in addition to a simple temperature mapping of the density \(\:\rho\:\left(T\right)=\frac{{\rho\:}_{ref}}{1+\alpha\:\left(T-{T}_{ref}\right)}\:,\) the viscoelastic behaviors affecting the system (i.e. thermal contraction and viscosity changes) with temperature may be parameterized as a function of \(\:{T}_{g}\). This enables us to simulate model “DMSO-like” systems with varying glass transition temperatures by rationally scaling the empirical temperature-dependent properties of water-DMSO to different input \(\:{T}_{g}\) values. Recapitulating specific aspects of our experimental setup, we include the initial cavity in the sample as a free-moving boundary, and fix the enclosing washer as a constrained non-contracting boundary. We apply a 100 °C/min cooling rate to the outside boundary of the washer, consistent with the approximate cooling rate observed at that position in preliminary experiments. While this approach is idealized, we argue that it yet captures the thermomechanical trends of relevance to the experiments presented herein. All supporting equations and properties for the simulation are detailed in Supplementary Note 1 (Supplementary Figures S1 and S2).
Under these conditions, the stress field is dominated by in-plane thermal contraction, with the geometry enforcing nearly axisymmetric deformation. As demonstrated in Supplementary Notes 6 and 7 (Supplementary Figures S9, S11), both the first and second principal stresses are tensile and nearly identical in magnitude and spatial distribution due to this 2D constraint, while the third principal stress remains low and compressive across all regions. Because fracture in vitrified systems is governed primarily by tensile failure18, and because the out-of-plane stresses contribute minimally to crack initiation, we focused our analysis on the first principal stress as the most relevant predictor of fracture risk.
Figure 4 provides the simulated effect of varying glass transition temperatures on the maximum (Fig. 4.A) and average (Fig. 4.B) first principal stress over the course of the vitrification process, and indicates that lower glass transition temperatures produce higher maximum and average first principal stresses (for the same cooling protocol). Consistent trends in stress development with temperature are observed across simulations, which vary in magnitude with system \(\:{T}_{g}\). At the point of maximum stress (Fig. 4.A), which, consistent with prior observation18, invariably occurs at the center-facing tip of the expanding cavity (Supplementary Note 2, Supplementary Figures S3 and S4) where surface deformation is maximal, the stress increases sharply upon transition to glass; enters a period of slower stress increase as the rest of the sample vitrifies; and finally moderately decreases upon thermal equilibration and relaxation. This same phenomenology is present in the average stress (Fig. 4.B), though distributed less sharply over time/temperature as expected.
The stress magnitudes of these responses are driven by multiple interacting factors. First, surface deformation occurring in the cavity regions during cooling of the contracting liquid becomes mechanical strain as the material transitions from a flowing viscous liquid into a non-flowing, brittle solid (Supplementary Note 2, Supplementary Figures S3 and S4). The extent of this deformation increases with decreasing glass transition temperature both because more total thermal contraction is allowed before the relative immobilization of the system at glass transition (i.e. the delta between room temperature and \(\:{T}_{g}\) is greater) and because, per the insights of Lunkenheimer and colleagues, the per-temperature rate of thermal contraction in the liquid is higher.
Second (and related), inadequate stress relaxation preceding the onset of the glass transition increases stresses throughout the system. Lower glass transition temperatures stymie stress relaxation (within a given cooling protocol) by way of both greater temperature gradients across the sample at the onset of glass transition and faster convective flow preceding glass transition, as evident in both the velocity plot shown in Fig. 4.C, which reports the speed of the deforming cavity-liquid interface where strain is maximum, and in the 2D velocity profiles shown in Supplementary Note 2, Supplementary Figures S3.C and S3.F. The temperature changes, thermal stress, velocity, and cavity deformation over time are presented in Supplementary Video 2.
In Fig. 4.D, we also show the development of the maximum stress post-glass transition as a function of cooling past the glass transition temperature. This data demonstrates that \(\:{T}_{g}\) substantially affects the stress profile before and after solidification; before by affecting the degree of surface deformation, and after by affecting the continued contraction of the glass (by way of the Lunkenheimer relation) and the relative stress relaxation of the entire system. We note that the maximum stresses observed in all simulations, which vary by a factor of approximately 4 from \(\:{T}_{g}\) = -80 °C to \(\:{T}_{g}\) = -130 °C, are all consistent with the brittle fracture observed across our experiments, which has been reported previously to occur at as low as 2–3 MPa33. In Supplementary Note 3 (Supplementary Figure S5), we also provide alternate simulations accounting for the contraction of the aluminum washer used in our experiments. Trends between samples, general stress magnitudes, and general phenomenology are preserved.
To further support the generalized parametric analysis performed thus far, in which we have simulated a model DMSO-like liquid with thermophysical properties scaled to accommodate a given enforced glass transition temperature, in Supplementary Note 6 we perform identical simulations incorporating empirical thermophysical properties from of each of the four solutions experimentally studied (Supplementary Figures S8, S9, Tables S5-S7). These include measured viscosities and specific heat capacities, alongside estimated thermal conductivity and density based on pure component data from the literature. Critically, when using these solution-specific thermophysical data, we observed precisely the same trends with Tg observed in the parametric simulations.
Additionally, to verify the implicitly assumed relationship between accumulated thermal stress and ultimate brittle fracture, we also implemented a coupled phase-field fracture model, as detailed in Supplementary Note 7. This approach simulates the evolution of a damage field during cooling, capturing crack nucleation and propagation without requiring predefined crack paths. The phase-field results confirm that the same Tg-dependent behavior observed in both our experiments and thermomechanical stress simulations is preserved: lower Tg mixtures exhibit greater damage accumulation due to increased thermal contraction and stress concentration. These results reinforce our conclusion that glass transition temperature is the dominant factor governing fracture risk during vitrification (Supplementary Figure S10).
We note that while the phase-field model incorporates fracture resistance through the critical energy release rate (Gc), we recognize that Gc is directly related to fracture toughness, and that both may also vary with Tg34. Although direct measurement of fracture toughness at cryogenic temperatures is currently limited by experimental constraints, we used literature-consistent estimates of Gc deemed appropriate for brittle vitrified materials near their Tg (Supplementary Note 7). Understanding how fracture toughness itself may correlate with Tg remains a promising area for future investigation. In Supplementary Note 8, we provide examples of this correlation from the bulk metallic glass literature, but note that this relationship has not to our knowledge been explored in the context of aqueous organic glasses.
Effect of solution thermal conductivity
Finally, we sought to analyze how varying heat transfer dynamics might affect these results. In order to maintain relevance to the absolute cooling rates employed in our experiments, we tune heat transfer within the simulated sample by varying sample thermal conductivity at a constant glass transition temperature (here − 130 °C).
Figure 5 shows how the maximum (Fig. 5.A, C) and average (Fig. 5.B) first principal stresses (dashed lines) vary with different scales of thermal conductivity, alongside the corresponding temperature profiles on the right axis (solid lines). Interestingly, and counter to intuition, lower thermal conductivities are observed to decrease thermomechanical stress, even while increasing the maximum thermal gradients observed during cooling (which are often taken generically to increase thermal stress9. This effect appears to be dominated by stress relaxation processes, with lower thermal conductivity leading to slower diffusion of heat out of the interior of the sample and, accordingly, greater time for structural equilibration. Likewise, higher thermal conductivities appear to drive a growing mismatch between the thermal and structural equilibration timescales, minimizing stress relaxation and maximizing peak stresses.
These observations appear qualitatively consistent with observations by Rabin that slower cooling rates decrease thermomechanical stresses driven by surface deformation in a cylindrical geometry18. However, slower cooling rates in that context are also accompanied by reduced thermal gradients; our observation herein thus clarifies that the effect of steep temperature gradients may be decoupled from the stress profile at a given cooling rate via the thermal conductivity, which here dictates the characteristic timescale available for stress relaxation. This interplay warrants further examination, and is likely highly dependent on system geometry (and especially the position of the deforming liquid-gas interface).
Thermomechanics analyses of first principal stresses in a model vitrification solution with a glass transition temperature of -130 °C and different assumed thermal conductivities. (a) Maximum first principal stress during cooling, sampled at the bottommost point of the liquid-cavity interface, where deformation is greatest. (b) Average first principal stress throughout the entire sample during cooling. (c) Maximum principal stress plotted as a function of difference in temperature (ΔT) from the glass transition temperature \(\:{T}_{g}\). In all panels, results are provided for samples with various thermal conductivities scaled relative to that of the reference solution, 7.05 M DMSO in water.
Discussion
Limitations of the present study
The collected experimental and computational results relayed herein suggest that the glass transition temperature of an aqueous solution may be a dominant predictor of thermal stress and fracture, based on a suite of multiphysics effects related ultimately to its inverse relationship with thermal expansion17. This suggestion is consistent with the sparse prior literature that has (indirectly) probed this phenomenon experimentally30, but our interpretation is subject to several key limitations.
Firstly, we have interpreted our cracking experiments in terms of the thermal stresses that create the necessary conditions for initial crack formation, rather than in terms of those material properties (toughness, ductility, etc.) that dictate the extent and style of crack propagation. Future studies must interrogate to what degree these latter properties vary amongst the solutions studied here, and to what degree they may affect the experimentally observed fracture outcomes. We note however that if fracture-related material properties of the glassy phase were dominating the experimental outcomes observed in Fig. 2, one may expect that materials with higher glass transition temperatures would produce more cracking, as they are cycled farther past their glass transitions when cooled to -196 °C, and deeper into the brittle regime.
Secondly, our computational analyses assume that the Lunkenheimer relation17 linking increasing \(\:{T}_{g}\) with decreasing thermal expansion is generically valid for the solutions studied herein, though aqueous organic solutions of this sort are only minimally represented in their dataset. While we assert that our experimental data indicates that their relation likely holds, a certain circularity of logic is introduced that can only be circumvented by further experimentation. Future experimental characterization of the thermal expansion of cryopreservative solutions of varying glass transition temperatures may elucidate the present findings. Furthermore, we note that system geometry plays a strong role in the generation of thermal stress, especially with regard to the geometry of free-moving deformable boundaries such as the liquid-air interface18,19, and that the extensibility of our present findings to arbitrary alternative geometries must be further examined.
Outlook and applications to cryobiology
This work in total suggests an underappreciated role of the glass transition temperature itself in determining thermomechanical outcomes during cryopreservation, and in doing so highlights the minimally explored chemical parameter space to which state-of-the-art physical analysis of vitrification cryopreservation has been applied. Significant recent work has highlighted the need for (and limited understanding of) new cryoprotectant chemistries35,36,37, motivated both by reducing toxicity and by improving thermodynamic outcomes. As these new chemistries enter the toolkit of the cryobiologist, opportunities abound to rethink classical conceptions of the necessary behaviors of vitrification solutions, which to date have been based on a small handful of often chemically-similar cryoprotectant molecules.
Our work herein suggests that design of solutions with higher glass transition temperatures may provide a valuable route by which to reduce the practical challenge of thermal stress cracking, which poses a significant barrier to volumetric-scale up of vitrification procedures. Higher glass transition temperatures also correspond generally to higher stability against crystallization en route to liquid nitrogen temperatures and a narrower temperature window in which the solution may be susceptible to ice nucleation7,37. Collectively, these factors suggest that incorporation of the glass transition temperature as a target variable in the design of next-generation vitrification solutions may enable circumvention of thermal stress failures and other landmark challenges in cryopreservation by vitrification.
Materials and methods
Given the focus of this work on the experimental platform and method itself, relevant methodological details are provided throughout the main text. Where additional details are necessary to facilitate reproduction, they are included below. A comprehensive description of our computational thermomechanics methods and image processing methods are included in the Supplementary Information.
Solution Preparation
All chemicals were sourced from Sigma Aldrich, and all solutions were prepared on a mass/mass basis in de-ionized water. Solutions were briefly ultrasonicated at room temperature prior to use to remove excess dissolved gases and errant air bubbles.
DSC measurements
The glass transition temperatures of the four binary solutions investigated were determined using two different methods, as described in Supplementary Note 4: differential scanning calorimetry (DSC) and constant light-source optical intensity tracking in a custom isochoric cryomacroscope platform, as described in24. The warming cycle of all acquired thermograms are shown in Supplementary Figure S6.A, and warming-cycle optical intensity curves are shown in Supplementary Figure S6.B. Glass transition values were extracted as described for both techniques in24, and are reported in Supplementary Table S2 alongside literature values. For the experimental comparisons described in the main text, we use the average of the values listed in Table S2. DSC measurements were conducted using a Q2000 series device from TA Instruments. 15 mg samples were transferred via micropipette to Tzero-brand aluminum hermetic DSC pans, which were then lidded and sealed using a Tzero press. The temperature was then cycled from room temperature to -150 °C at cooling and warming rates of 5 °C/min. Onset glass transition temperatures were extracted in accordance with standard ASTM E1356-08.
Computational analyses
Comprehensive descriptions of our thermomechanics computations and our image processing computations are provided in Supplementary Note 1 (Supplementary Figures S1, S2, Table S1) and Supplementary Note 5 (Supplementary Figure S7, Tables S3 and S4) respectively.
Statistical analysis
The statistical analysis shown in Fig. 3 was conducted using the anova1() function in MATLAB 2023a, with a p > 0.05. defining the threshold for statistical significance.
Data availability
All data shown herein is available upon reasonable request to corresponding author Matthew J. Powell-Palm (powellpalm@tamu.edu).
Code availability
All MATLAB scripts used for the research herein are available upon reasonable request to the authors.
References
Giwa, S. et al. The promise of organ and tissue preservation to transform medicine. Nat. Biotechnol. 35, 530–542 (2017).
Han, Z. et al. Vitrification and nanowarming enable long-term organ cryopreservation and life-sustaining kidney transplantation in a rat model. Nat. Commun. 14, 3407–3407 (2023).
Powell-Palm, M. J. et al. Cryopreservation and revival of Hawaiian stony corals using isochoric vitrification. Nat. Commun. 14, 4859–4859 (2023).
Hagedorn, M. et al. Safeguarding earth’s biodiversity by creating a lunar biorepository. Bioscience 74, 561–566 (2024).
Deck, L. T. et al. Monitoring aqueous sucrose solutions using droplet microfluidics: ice nucleation, growth, glass transition, and melting. Langmuir 40, 6304–6316 (2024).
Deck, L. T., Gusev, N., Deligianni, V. & Mazzotti, M. Quantifying the effect of particulate impurities on the ice nucleation behavior of pharmaceutical solutions. Int J. Pharm., 125137 (2025).
Fahy, G. M. & Wowk, B. Principles Cryopreservation Vitrification pp. 21–82. (2015).
Fahy, G. M., Saur, J. & Williams, R. J. Physical problems with the vitrification of large biological systems. Cryobiology 27, 492–510 (1990).
Steif, P. S., Palastro, M. C. & Rabin, Y. The effect of temperature gradients on stress development during cryopreservation via vitrification. Cell. Preserv. Technol. 5, 104–115 (2007).
Steif, P. S., Palastro, M. C. & Rabin, Y. Analysis of the effect of partial vitrification on stress development in cryopreserved blood vessels. Med. Eng. Phys. 29, 661–670 (2007).
Solanki, P. K. & Rabin, Y. Thermomechanical stress analysis of rabbit kidney and human kidney during cryopreservation by vitrification with the application of radiofrequency heating. Cryobiology 100, 180–192 (2021).
Eisenberg, D. P., Bischof, J. C. & Rabin, Y. Thermomechanical stress in cryopreservation via vitrification with nanoparticle heating as a stress-moderating effect. J. Biomech. Eng. 138, 011010 (2016).
Gao, Z. et al. J. C. Bischof, vitrification and rewarming of magnetic Nanoparticle-Loaded rat hearts. Adv Mater. Technol 7, (2022).
Rabin, Y. In Thermomechanics Modeling and Visualization of Physical Effects To Improve Cryopreservation by Vitrification in Annual Review of Heat Transfer. Vol. 27, 245–282 (eds Bischof, J. C.) (Begell House Inc., 2024).
Manuchehrabadi, N. et al. Improved tissue cryopreservation using inductive heating of magnetic nanoparticles. Sci. Transl. Med. 9, eaah4586 (2017).
Han, Z. et al. Supplemented phase diagrams for vitrification CPA cocktails: DP6, VS55 and M22. Cryobiology 106, 113–121 (2022).
Lunkenheimer, P., Loidl, A., Riechers, B., Zaccone, A. & Samwer, K. Thermal expansion and the glass transition. Nat. Phys. 19, 694–699 (2023).
Rabin, Y. Mathematical modeling of surface deformation during vitrification. Cryobiology 102, 34–41 (2021).
Vispute, D. M., Solanki, P. K. & Rabin, Y. Large surface deformation due to thermomechanical effects during cryopreservation by vitrification – mathematical model and experimental validation. PLoS ONE 18, (2023).
Shaw, J. M., Kuleshova, L., Macfarlane, D. R. & Trounson, A. O. Vitrification properties of solutions of ethylene glycol in saline containing PVP, ficoll, or dextran. Cryobiology 35, 219–229 (1997).
Fahy, G. M. & Wowk, B. Principles of cryopreservation by vitrification. Cryopreservation Freeze-drying Protocols, 21–82 (2015).
Rasmussen, D. & MacKenzie, A. Phase diagram for the system water–dimethylsulphoxide. Nature 220, 1315–1317 (1968).
Zamecnik, J., Faltus, M. & Bilavcik, A. Vitrification solutions for plant cryopreservation: modification and properties. Plants 10, 2623 (2021).
Kavian, S. et al. Powell-Palm, an isochoric optical platform for interrogation of aqueous glass formation processes. RSC Adv. 14, 34594–34605 (2024).
Brostow, G. J., Fauqueur, J. & Cipolla, R. Semantic object classes in video: A high-definition ground truth database. Pattern Recognit. Lett. 30, 88–97 (2009).
Chen, L. C., Zhu, Y., Papandreou, G., Schroff, F. & Adam, H. In Proceedings of the European conference on computer vision (ECCV). pp. 801–818. (2018).
Cook, R. F. Thermal activation effects in crack propagation and reliability of fused silica. J. Am. Ceram. Soc. 102, 7575–7583 (2019).
Schoeck, G. Thermally activated crack-propagation in brittle materials. Int. J. Fract. 44, 1–14 (1990).
Kuleshova, L. L., MacFarlane, D. R., Trounson, A. O. & Shaw, J. M. Sugars exert a major influence on the vitrification properties of ethylene glycol-based solutions and have low toxicity to embryos and oocytes. Cryobiology 38, 119–130 (1999).
Shaw, J. M., Kuleshova, L. L., MacFarlane, D. R. & Trounson, A. O. Vitrification properties of solutions of ethylene glycol in saline containing PVP, ficoll, or dextran. Cryobiology 35, 219–229 (1997).
Pryor, R. W. Multiphysics modeling using COMSOL®: a first principles approach (Jones & Bartlett, 2009).
Trejo Gonzalez, J. A., Longinotti, M. P. & Corti, H. R. The viscosity of glycerol – water mixtures including the supercooled region. J. Chem. Eng. Data. 56, 1397–1406 (2011).
Rabin, Y., Steif, P. S., Hess, K. C., Jimenez-Rios, J. L. & Palastro, M. C. Fracture formation in vitrified thin films of cryoprotectants. Cryobiology 53, 75–95 (2006).
Anderson, T. L. & Anderson, T. L. Fracture Mechanics: Fundamentals and Applications (CRC, 2005).
Warner, R. M. et al. Rapid quantification of multi-cryoprotectant toxicity using an automated liquid handling method. Cryobiology 98, 219–232 (2021).
Warner, R. M., Brown, K. S., Benson, J. D., Eroglu, A. & Higgins, A. Z. Multiple cryoprotectant toxicity model for vitrification solution optimization. Cryobiology 108, 1–9 (2022).
Murray, K. A. & Gibson, M. I. Chemical approaches to cryopreservation. Nat. Reviews Chem. 6, 579–593 (2022).
Funding
Funding is gratefully acknowledged from the NSF Engineering Research Center for Advanced Technologies for Preservation of Biological Systems (ATP-Bio) under Grant No. EEC 1941543. The authors also thank Dr. Anthony Consiglio and Dr. Arun Srinivasa for helpful discussions.
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Kavian, S., Sellers, R., Sanchez, G.A. et al. Higher glass transition temperatures reduce thermal stress cracking in aqueous solutions relevant to cryopreservation. Sci Rep 15, 27903 (2025). https://doi.org/10.1038/s41598-025-13295-7
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-13295-7
Keywords
Supplementary Material 1 Supplementary Material 2