- Article
- Open access
- Published:
Nature Communications volume 17, Article number: 3618 (2026) Cite this article
-
2726 Accesses
-
129 Altmetric
Subjects
Abstract
Despite coffee’s popularity, there are no quantitative methods to measure a chemical property of a black coffee drink in situ and relate it to a flavor experience. Here we show that cyclic voltammetry can be used without any additional sample preparation to directly measure the strength of a coffee beverage and, separately, how dark the coffee has been roasted; these two properties are implicated in the sensory profile of the beverage. We show that the current passed for the cathodic features that precede hydrogen evolution are linearly related to beverage strength. The same features are suppressed with subsequent cycling due to coffee material accumulating on the electrode. The magnitude of suppression is directly related to roast color, which dictates the ensemble chemical composition and flavor of the beverage. Together, this voltammetric analysis decouples beverage strength from roast color and offers a strategy for rapidly assessing flavor-correlated chemical properties of coffee.
Similar content being viewed by others
Introduction
Since the 1950s, the coffee industry has sought quantitative methods to assess beverage qualities beyond those informed by sensory panels. In the meantime, a litany of research on the topic has revealed that beverage concentration1,2, and roasted bean color3,4, are the two primary and independent factors that dictate the sensory perception of coffee. Bean color is readily determined by spectrophotometry5,6,7, while the most widely used technique to measure concentration of solvated coffee relates the refractive index of the beverage8,9, to an effective concentration through an empirically derived polynomial. The refractive index method reports an approximated total dissolved solids (wt.% TDS) ratio of the mass of dissolved coffee to the mass of the beverage and informs beverage strength10. From wt.% TDS, the efficiency of the extraction process can then be calculated by taking the mass ratio of the dissolved solids to the dose of ground coffee (i.e., extraction yield11). The industry has coalesced around the phenomenological observation that dissolution of ≈20% of the dry mass, yielding a beverage ≈1.35 wt.% TDS generally produces an enjoyable cup of filter coffee12,13. To date, measurements based on refractive index remain the industry standard. However, the refractive indices of solutions are dependent on the identity of the solutes. Glucose, for example, will have the same refractive index at 2% w/w as a solution of ethanol that is twice as concentrated, making refractive index measurements unable to identify chemical differences in more complex mixtures (e.g., coffee), and particularly compositions result in dissimilar sensory outcomes14,15,16,17,18,19. Given the composition of roasted coffee primarily depends on roast color, the existing quantitative approach cannot discern differences between light-roasted and dark-roasted coffees that achieve the same refractive index, let alone higher-fidelity chemical differences achieved in the same coffee using modified brewing parameters or roast profiles. A rapid quantitative method that is sensitive to compositional information beyond wt.% TDS remains a major target for the industry.
There exist numerous analytical techniques that provide both qualitative and quantitative information about chemical composition, with the gold standard being liquid or gas chromatography coupled with mass spectrometry for soluble and volatile compounds, respectively. Besides the obvious challenge of identifying specific compounds among the thousands observed in coffee that give rise to a measurable sensory experience, these techniques also suffer from slow run times, laborious sample preparation, and high associated costs while yielding limited predictive insights. Instead, some research groups have used electrochemical techniques to measure the concentrations of common molecules in solution20,21. Electroanalytical techniques measure the amount of current passed between electrodes immersed in the solution at voltages where solvated molecules undergo redox reactions; the measured current is directly proportional to the local concentration of the reacting molecule(s). While this approach is generally sensitive enough to accurately measure the concentration of caffeine22, various chlorogenic acids (CGAs)23, and other molecules24, previous reports have omitted beverage strength in their analyses, precluding the development of a technique which relates preparation variables (e.g., mass of coffee, mass of water, grind setting, water temperature and pressure, contact time, roast color etc.) to the resultant chemical composition of the liquid.
Herein, we report an advance in coffee quality analysis that harnesses in situ changes in the protonic electrochemical response (e.g., hydrogen underpotential deposition, HUPD) in cyclic voltammetry (CV) measurements of liquid coffee. By sampling features in the electrochemical response that are affected by the ensemble chemical composition of the coffee rather than measuring the concentrations of individual molecules, this approach captures quantitative information about both roast color and beverage strength. These two properties drive the sensory profile of the beverage, thereby allowing this analytical technique to exceed the insights provided by refractive index measurements and provide additional quality information that correlate with flavor.
Results
Initially, we sought to directly measure the concentration of caffeine (Eox = ≈1.4 V vs. Ag/AgCl25), CGAs (Eox = ≈0.2–0.5 V vs. Ag/AgCl, depending on the isomer23), and other redox-active species in undiluted coffee samples to study their dependence on conventional brew parameters. However, caffeine and CGAs form an aggregate at concentrations typical of brewed filter coffee26, terminally impacting their redox activity—they only become electrochemically well-resolved in acidified dilute solutions with added electrolyte. Literature examples dilute to below 0.02 wt.% TDS27, nearly two orders of magnitude weaker than filter coffee. Other groups have shown that by adulterating coffee with either CGA or caffeine, both boron-doped diamond (BDD) and glassy carbon (GC) electrodes can provide molecule-specific information28,29,30,31,32,33,34,35,36. Yet, these approaches required laborious sample preparations, in opposition of both our and the industry's goal of measuring features of as-consumed coffee. Ideally, ensemble chemical measureables map to sensory experiences and relate the electrochemical features to total beverage strength, rather than measuring the concentration of specific molecules contained within. Thus, we first ascertained the redox landscape in filter-strength coffee by making CV measurements using BDD, GC, and Pt working electrodes across the electrochemical window of the beverage (Fig. 1a).
a Cyclic voltammetry performed at 200 mV s−1 using boron-doped diamond, glassy carbon, and platinum working electrodes. Caffeine and chlorogenic acid potential ranges studied by other groups are highlighted. Here, we focus on the protonic features (HUPD and acid redox) region. b These features are suppressed with subsequent CV cycling due to deposition of coffee material on the surface of the electrode, and the total charge can be extracted by subtracting the hydrogen evolution reaction (HER) background. c The current depends on wt.% TDS of the brewed coffee because the protonic and organic concentration scales with wt.% TDS. d Scanning anodically results in mass accumulation on the working electrode, leading to electrode fouling. e The mechanism of mass accumulation is likely proton-assisted, given that appreciable mass does not deposit until the surface has accumulated a critical H-atom concentration. Source data are provided as a Source data file.
As-consumed filter coffee extracts are sufficiently conductive for direct electrochemical analysis without the addition of a supporting electrolyte (ranging from 2-3 mS cm−1, see Supplementary Fig. 1). Additionally, coffee extracts are self-buffered to pH of ≈4.8–5.9 depending on the distribution of compounds in the coffee and brew water composition37,38,39. Even after performing bulk electrolysis at oxygen evolution potentials for two minutes, the pH of a typical filter coffee remains numerically identical, reinforcing the significant buffering capacity of brewed coffee. Yet, despite the plethora of molecules in coffee extracts, the CV response of a Pt working electrode in 1.56 wt.% TDS coffee is consistent with that of dirty acidic water (Fig. 1a)40,41,42,43,44,45.
The cathodic Faradaic features map to the response expected for protonic reactions with the Pt surface (e.g., HUPD), followed by H2 evolution at more negative potentials. At positive potentials, OH adsorption and eventual O2 evolution are also evident. To ascertain whether the anodic feature at –0.6 V is linked with the cathodic features in a reversible redox couple, we probed the scan rate dependence, Supplementary Fig. 2. The linear dependence of peak current on the square root of the scan rate for both features demonstrates the diffusion-controlled nature of the redox events, Supplementary Fig. 3, and the lack of an increase in the peak potential separation with increasing scan rate indicates Nernstian behavior, Supplementary Fig. 4. However, the large peak separation of ≈200 mV at all scan rates suggests that the reversibility of the redox couple is obfuscated by sluggish kinetics.
The same Pt surface sites that adsorb H+ and OH– are also able to adsorb other molecules in solution. In the case of oxidative cycling, some impurities in water compete for the Pt surface, resulting in reduced current with subsequent cycling due to a decrease in the accessible surface area. Given that Pt is known to interact with caffeine and other molecules in coffee46,47, we expected to see a decrease in exchange current density with sequential cycling. When scanning from 0 to –1.0 V, the HUPD and protonic features (Epc = –0.4 and –0.7 V, respectively) smear together and current decreases by ≈34% from CV scan 1 to 2 and ≈18% from scan 2 to 3 (Fig. 1b)48. In pH 7 water purified by reverse osmosis, the same features are not observed (Supplementary Fig. 5), suggesting that the response is due to protonic chemical steps associated with the coffee and not the water.
Further experiments were run to ensure that these features mapped to HUPD/weak acid reduction and its suppression by coffee molecules, rather than fluctuations in dissolved O2 and other spurious effects, Supplementary Fig. 6. Since the integral of the current density depends on the activity of H+, the HUPD and acid features indirectly provide ensemble insights into the families of molecules in coffee that function as H+ donors and acceptors, the concentrations of which should depend on roast color, brewing parameters, brew water composition, and so forth. Some data in support of this hypothesis is that HUPD/acid reduction current density decreases with decreasing coffee concentration due to the diminished concentration of available H+ (Fig. 1c). Because the feature is concentration dependent, there are also fewer organic molecules competing to bind to the surface of the electrode. As we will show later, the integral of the charge current density of this feature linearly maps to wt.% TDS. Perhaps this is a surprising result, given that a single acidic feature should not necessarily depend on ensemble concentration.
To further support our proposed mechanism that protonic chemistry is being suppressed by adsorbed coffee material, we performed CV measurements using an electrochemical quartz crystal microbalance (QCM) with a Pt working electrode scanning at 50 mV s−1 (Fig. 1d). Scanning cathodically, appreciable mass begins to accumulate on the electrode at potentials more negative than HUPD—the balance is insensitive to surface H-adsorption but can detect larger molecule accumulation. There is a delayed onset, which attribute to a proton-assisted adsorption of Brønsted-basic species like caffeine, following a general mechanism (Fig. 1e). Upon scanning anodically from −1.2 to 0.0 V, mass continues to accumulate until the potential exceeds −0.5 V, when the electrode liberates most of the adsorbed organic material and protons back into solution. The response is reminiscent of a kinetic trap, where the surface assembly forms in kinetically favored conditions49. To ensure that the process was proton assisted, we also scanned from −1.2 to 0.0 V without first scanning cathodically and detected no mass accumulation, indicating that there must be an appreciable concentration of protons on the surface before larger organic species begin to accumulate, Supplementary Fig. 7.
While the total charge passed maps linearly to concentration for any particular coffee, coffees from different origins, processed in dissimilar ways, and roasted to different colors may show major differences in the emergence and suppression of the reductive features. However, before we can probe coffee-related variables we must first ensure that the suppression of the convoluted redox feature at −0.55 V depends on molecules likely found in all coffees. To determine the molecular identities of the adsorbed compounds, we developed the CV cycle shown in (Fig. 2a) to maximize mass accumulation on the Pt working electrode for further characterization. We were able to obtain sufficient coffee material on a Pt-mesh electrode surface by cycling 100 times from −0.55 to −1.2 V (Fig. 2a), followed by solvating the adsorbed material in a sonicated 4 mL bath of 80/20 water/acetonitrile (v/v), and repeating four times. The adsorbates could then be separated and characterized using high performance liquid chromatography coupled with high-resolution mass spectrometry (HPLC-MS, Fig. 2b). We found that caffeine had adsorbed in quantifiable concentration. Our combined samples yielded 7.8 ± 0.1 mg kg−1 caffeine, suggesting that at least one component of the accumulated mass that causes the current to decay over successive CV cycles originates from a molecule common to all coffees, and that our 4 mL solution contained ≈300 µg of caffeine, or ≈0.4% of the total caffeine in an average 180 mL cup of filter coffee50,51,52, (see Supplementary Figs. 8,9). That is, each 100-cycle CV presented in (Fig. 2a) scavenged ≈0.1% of the available caffeine in the cup, as well as other molecules.
a Cyclic voltammetry performed at 200 mV s−1 sampling potentials more negative than –0.5 V, to ensure our accumulated mass is maximized rather than liberated back into solution (Fig. 1d). The first four cycles of the second run are presented. b Combining the extracts from the surface of the Pt electrode sampled 100 times in four separate runs, we can detect the presence of caffeine and quantify it using the calibration curve presented in Supplementary Fig. 8. c The adsorption of caffeine and 5-caffeoylquinic acid is favored on all clear crystal facets of Pt according to density functional theory simulations, suggesting that a collection of organic molecules responsive to roast color and brewing parameters adsorb to the electrode surface. Source data are provided as a Source data file.
Given that a similar electrochemical approach has been used to quantify caffeine content in highly dilute coffee samples through oxidation29, and that bulk Pt also is known to adsorb organic material40, we were somewhat unsurprised to see caffeine in the chromatogram. However, there are of course some coffees that have been decaffeinated, prompting us to resolve whether this mass deposition can be attributed to additional adsorbates beyond caffeine. Because other adsorbates were not in sufficient concentration to quantify with HPLC-MS, we instead turned to density functional theory (DFT) paired with molecular dynamics simulations (MD) to model the Pt surface adsorption energies for caffeine as well as 5-caffeoylquinic acid (5-CQA), an abundant CGA isomer found in all coffees with a concentration that depends strongly on roast parameters50,53,54, (Fig. 2c). The inclusion of caffeine in the DFT study serves as a control to validate the model, and the inclusion of 3 low-index Pt surfaces, (100), (110), and (111), examines the possibility of preferential binding to certain facets of the polycrystalline Pt electrode used for the electrochemical measurements. The model we use does not account for the significant thermodynamic parameter of solvation, as explicit treatments of solvent molecules incur high computational costs and implicit models often fail to reproduce experimental solution-state adsorption energies. Because the penalty of excluding water from the Pt surface upon adsorption is unaccounted for, the calculated adsorption energies likely overestimate the attractive force of the interaction55. Furthermore, the exclusion of an applied electric field in the DFT model precludes both the assertion of equilibrium geometries for the Pt/adsorbate complex at potentials corresponding to electrochemical processes and the calculation of accurate thermochemical parameters for adsorbate formation. Nevertheless, a model of the electrode/adsorbate interface at zero field is instructive for determining relative trends of adsorption between similar molecules that form similar surface dipoles, and for assessing the existence or lack of facet dependence in the complex56.
For each combination of molecule and surface, we obtained several local-minimum configurations from the MD trajectories and then ran DFT structural optimizations to obtain equilibrium energies; the difference in energy between the molecule-surface complex and the isolated molecule and surface defines the adsorption energy, with a more negative value representing stronger binding. The adsorption of caffeine to three facets of Pt revealed that each facet stably binds caffeine as expected, with a slight preference for the (110) surface. Our calculations reveal that 5-CQA also forms stable complexes with the three Pt surfaces, albeit with a weaker interaction than caffeine, and prefers the (111) surface. Interestingly, 5-CQA binds least strongly to the (110) surface, indicating that specific crystal facets of the Pt electrode may offer some degree of selectivity for adsorbed species, which in turn may prove useful in follow-up work that harnesses the kinetics of adsorption to parse flavors. Together, the DFT models instruct that the suppression of the HUPD and protonic features likely captures the ensemble of various organic adsorbates binding to the electrode surface, providing information about the beverage concentration, as well as roast color, since roast will dictate the amount of 5-CQA (and caffeine57) in the cup.
Since the Faradaic protonic features depend on coffee concentration, we sought to elucidate how roast color (and subsequent chemical composition) impacts the redox response. To do so, we roasted a representative specialty coffee sourced from Colombia, following the profiles shown in Fig. 3a. The “light roast” profile was then systematically extended by 30 s and in final temperature by 1.11 °C (2 °F), to achieve four progressively darker coffees. A final sample was prepared by extending the fifth roast by a further 60 s and 2.22 °C (4 °F) to ensure that the roast colors covered a broad and reasonable range of Agtron values, see Methods. The roast profiles yielded coffees ranging from 75.8 (light) to 55.7 Agtron units (dark). Each roasted sample was rested for 7 days to allow for CO2 off-gassing58 and then brewed using the Specialty Coffee Association cupping protocol59.
a The measured temperature profiles used to generate six systematically darker roasts. b The first cycle CV response of these coffees brewed and diluted to 1.00 ± 0.02 wt.% TDS. c Subtracting the background contribution due to the onset of hydrogen evolution Supplementary Fig. 10, the total charge passed is linear with wt.% TDS with a slope dependent on roast color. Darker roasts more effectively suppress the cathodic features, likely because they contain more solvated oxidized molecules formed during roast. These compounds evidently tend to bind more strongly to Pt. d Plotting the total charge and wt.% TDS against the ground coffee Agtron value (roast color), a 3D planar relationship is recovered. Error bars (±0.01 wt.% TDS, ± 0.1 × 10–5 C) for panel c are omitted for clarity but can be found in Supplementary Fig. 11. Source data are provided as a Source data file.
To isolate roast dependence, some sample preparation is required. By diluting the extracts to 1.00 ± 0.02 wt.% TDS, electrochemical differences that depend on roast color were noted in the voltammograms (Fig. 3b). The lightest-roast coffee passes ≈50% more charge than the darker analogues at the same refractive index-derived strength. While wt.% TDS remains fixed, the electrochemical assessment of the cathodic feature depends on roast, a critical parameter that dictates the sensory experience (i.e., dark-roasted coffees taste “dark”). By integrating the exchange current density and adjusting for scan rate, we can extract the total charge passed for each coffee at any arbitrary concentration (Fig. 3c). Here, each roast shows a strong linear fit between wt.% TDS and electrochemical charge passed. Increasingly darker roasts yield both a shallower gradient and less total charge passed than the lighter-roast counterparts. The diminished current may be expected from the fact that darker coffees tend to have mildly elevated pH60,61, and that dark roasts have less water-soluble material contained within, thereby preferentially depositing organic material onto the electrode. The rate of suppression of these redox waves depends on composition (see Supplementary Figs. 12–17). Together, we can conclude that while the feature is indirectly related to coffee composition, it is sensitive to differences in roast-derived species.
The wt.% TDS can then be plotted along with the coffee Agtron color (both whole bean and ground show the same trend, see Supplementary Fig. 23) and total charge passed in the HUPD region to yield a plane of values of integrated charge for all combinations of wt.% TDS and Agtron color (Fig. 3d). While the shape of the plane may be coffee-specific, the fact that roast color maps linearly to charge allows for a wealth of applications. For example, a series of simple CV measurements on progressively more dilute coffee will allow a roaster to rapidly construct a quality control calibration curve, enabling quantitative comparisons of separate batches of the same coffee roasted to the same color. In a roastery quality control setting, it is possible that batches of roasted coffee could have similar whole bean colors but different flavor profiles, which may be detectable electrochemically as well as on the cupping table.
As a demonstration of the analytical capabilities of this approach, we sourced four batches of the same coffee roasted to a target same whole-bean Agtron color (93.0 ± 1.0 Agtron units) from a specialty roaster (Colonna, Bath, UK). Among those four batches, one was rejected by the roaster because it did not meet the color tolerance (the coffee was too light, 98.9 Agtron units), and it consequently exhibited undesirable flavors during their sensory quality control. We wondered whether we could discern an electrochemical difference between the accepted and rejected samples, in essence performing at least as well as the human tongue in the roastery.
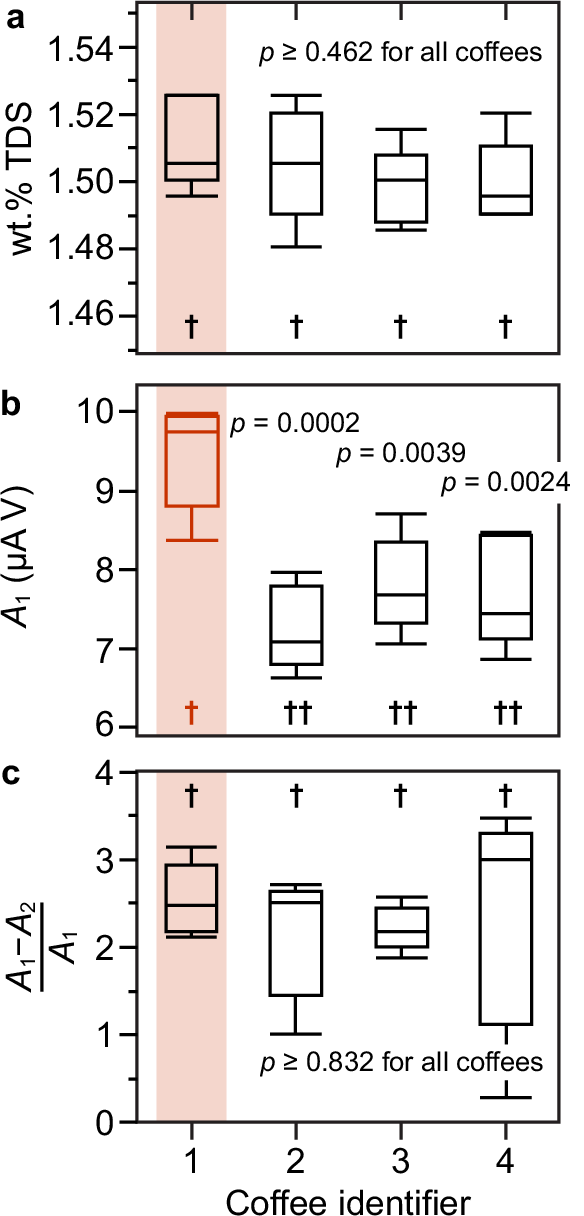
The roaster provided single-blind samples to us, labeled 1, 2, 3, and 4, and did not specify which was rejected. We prepared each sample in quintuplicate in a randomized order and brewed them according to the Specialty Coffee Association cupping technique. The samples were undiluted to allow wt.% TDS to vary, thereby introducing realistic error to our samples. After performing CV measurements on the samples in yet another randomized order, statistical analysis was performed using Tukey’s honest significant difference analysis of variance. The analysis revealed that the measured wt.% TDS of each coffee was statistically identical (Fig. 4); neither color analysis nor refractive index of the extracts could tell these coffees apart. However, examination of the current passed in scan one revealed that sample 1 was statistically dissimilar to the other coffees (p < 0.0039). The same was found for scans two and three. However, examining the difference in current passed between scan one and two, normalized by the initial current in scan one (i.e., (A1–A2)/A1), revealed that the rate of fouling of the electrodes was the same for all coffees (p > 0.832). That is, the rate of electrode fouling depends on concentration, but the charge passed depends on composition. By consulting with the roaster, sample 1 was revealed to be the rejected batch. Furthermore, the acceptable batches all fell within the same statistical class, demonstrating alignment between this electrochemical analysis and sensory profiling.
A single-blind assessment of four coffees roasted to the same specification, but one (sample 1) exhibiting negative flavors due to minor variation in roast parameters. Tukey’s honestly significant difference (HSD) ANOVA results reveal the measured wt.% TDS was statistically identical across all four samples (connecting letters † class); a wt.% TDS differences (insignificant) ranged from 0.002 ± 0.02 to 0.03 ± 0.02, with p = 0.999 to p = 0.462, respectively (n = 5). b The background-subtracted integrated cathodic area at –0.6 V for the first cycle, A1, was split into two distinct classes, † and ††, with sample 1 in class † and samples 2, 3, and 4 in class ††. c The differences between classes † and †† were 2.19 ± 0.40 µA V (1-2; p = 0.0002), 1.73 ± 0.40 µA V (1-4; p = 0.0024), and 1.64 ± 0.40 µA V (1-3; p = 0.0039). Differences between batches in the †† class (insignificant) ranged from 0.09 to 0.55 µA V (p = 0.996 to p = 0.529, respectively). The fouling was captured by normalizing the differences in charge passed between scans one and two, where all four samples were found to exhibit indistinguishable decreases in current with subsequent scans. A comprehensive comparison is presented in Supplementary Figs. 21 and 22. Box plots display the interquartile range (IQR), with whiskers indicating the minimum and maximum observed response values. Differences among treatments were evaluated using a two-sided comparison of means based on the studentized range statistic (q). Source data are provided as a Source data file.
Considering the findings in this paper, there is obvious upside to electrochemical assays of coffee extracts. Coffees can now be quality controlled and distinguished by their charge response—which is linked to the collection of molecules competing for the Pt surface—rather than the ensemble effect on refractive index. Given that we know that the response depends on the concentration of H+ (correlated with perceived acidity)62 but is suppressed by roast-dependent molecules in coffee like caffeine (bitter)63 and chlorogenic acid (astringent64, sour65), the electrochemical technique implicitly provides insights about flavor. Finally, this approach is sensitive to coffee composition and hence may find use in achieving desirable blends, detecting differences in seasonal crops, and resolving other high-fidelity coffee variables.
In conclusion, we have demonstrated that CV measurements performed in the cathodic region of the water window of brewed coffee produce a response aligned with the deposition of H+ on the Pt surface before forming H2. Since the charge passed during HUPD depends on the number of available surface sites on the Pt electrode, which is a function of the ensemble organic composition of the solution as well as its pH, these electrochemical features serve as a highly useful and sensitive measure of ensemble chemical composition in coffee samples. By performing a series of careful experiments, we show that the Pt surface also accumulates coffee material, leading to a reduction in charge passed with subsequent cycling, and identify that at least some fraction of this mass is associated with caffeine and other molecules in coffee, but not the brewing water itself. DFT simulations further instruct that the adsorption should generally have some dependence on molecular composition and reveal inhomogeneity in the solid-molecule interactions, and hence different coffees should suppress the cathodic feature to different extents. The most straightforward method to assess this was to simply prepare different roast levels of the same coffee. We demonstrated that in that series, coffees exhibit a linear relationship between beverage strength and total charge passed in the HUPD region, and that the rate of charge suppression with change in wt.% TDS depends on roast color. However, the technique is even more sensitive, as validated by matching human sensory discrimination (performed at a roastery during their quality control assessment). Our measurements correctly assign statistical classifiers to acceptable and unacceptable batches of roasted coffee with the same physical attributes. This suggests that the electrochemical method is sensitive to composition and together offers a reliable method to measure a critical aspect of coffee composition, as well as beverage strength, simultaneously.
Methods
Sample preparation
Green coffee was roasted in-house as described in the Roasting section. These roasts canvassed industrially relevant light, medium, and dark roasts (spanning whole bean Agtron values of 75.8 (lightest) to 55.7 (darkest), Supplementary Table 1). The coffee was allowed to rest for 7 days before being ground with a Mahlkönig EK-43 grinder (particle size distributions are presented in Supplementary Fig. 18) and brewed via cupping method with a brew ratio of 1:13.5 using 93 °C water (filtered Eugene tap water using a Pentair Everpure Conserv 75E Reverse Osmosis system with remineralization to 30 mg L−1 CaCO3). The coffee was allowed to contact the water for 4 min, without agitation or stirring. The samples were then filtered through a V60 filter paper to arrest brewing and allowed to come to room temperature (21.6 °C). The samples were stirred before each aliquot was taken to ensure homogeneity. The cumulative concentration of solvated coffee material (total dissolved solids as a mass fraction) was calculated from the measured refractive index following a procedure of ref. 66.
Electrochemical measurements
A three-electrode electrochemical setup was employed for all voltammetric measurements. CVs were performed using a Gamry 1010B potentiostat controlling Pt disk working (surface area, 0.0314 cm2) and Pt wire counter electrodes, and an Ag/AgCl (sat. KCl) reference electrode, purchased from CH Instruments. Prior to data collection, the working and counter electrodes were polished using a modified approach of ref.67 (10 scans at 300 mV s−1 from −0.3 to 1.0 V in 50 mM H2SO4, until key H2SO4 redox features overlaid, Supplementary Fig. 19). Signal-to-noise was then assessed through comparison between the brew water, the cleaning solution, and the coffee samples, Supplementary Fig. 20. Measurements were made directly on as-brewed coffee, with no supporting electrolyte or buffer. The solution-specific conductance of the coffee was measured using a Fisher Scientific Traceable conductivity probe calibrated with standard saturated solutions of KCl. The pH of each solution was noted in Supplementary Table 1. Since most coffee is a solution of weak acids, the solutions are self-buffering to a pH of around ≈5.0, aligned with ref. 38.
Particle size distribution
Particles created by the EK-43 were measured using a Malvern Mastersizer 2000 laser diffraction system. The solid samples were dispersed into an airstream (2.0 bar) of compressed breathing-quality air and passed through a He-Ne laser at 633 nm and solid-state blue light at 466 nm. Ground coffee samples were run in triplicate and the average particle size distributions are plotted in Supplementary Fig. 18.
Quartz crystal microbalance experiments
A QCM200 Quartz Crystal Microbalance equipped with a crystal holder was used to perform all QCM experiments. Pt-coated liquid plating 5 MHz quartz crystal electrodes with a Ti adhesion layer from Fil-Tech were used. The crystal holder ensures that only one side of the Pt-coated electrode is accessible to the solution, and the controller reads the associated frequency. A Biologic SP-300 potentiostat with a Pt wire counter electrode and Ag/AgCl (sat. KCl) reference electrode was used for collecting CV measurements corresponding with QCM data. The QCM frequency data were converted to mass values via the Sauerbrey equation as follows:
$$\triangle f=\frac{-{C}_{{{\rm{f}}}}\triangle m}{{{\rm{SA}}}}$$
(1)
\(f\) is the frequency as read from the QCM controller Cf, is the calibration constant, which is given to be 56.6 Hz cm2 μg−1 for the Fil-Tech electrodes, and SA is the surface area of the electrode, which is 0.4987 cm2.
High-resolution mass spectrometry
To accumulate mass on the electrode surface for further analysis, a Pt mesh electrode from BASi Research Products with a surface area of ≈5 cm2 was used as the working electrode with a Pt wire counter electrode and an Ag/AgCl (sat. KCl) reference electrode. The CV scan window was shortened to avoid the oxidation wave where most of the accumulated mass desorbed from the electrode surface. CV measurements were run in as-brewed coffee with about 1.5 wt.% TDS for 50-100 cycles. Immediately upon CV cycling completion, the working electrode was removed from the sample, patted dry with a Kimwipe, and submerged into a solution of 80% 18 MΩ cm water and 20% HPLC grade acetonitrile (Sigma-Aldrich) (v/v). The solution and working electrode were sonicated for 10 min and then the electrode was removed and rinsed. This process was repeated a total of four times to ensure that the solution contained enough redissolved residue for mass spectrometry analysis.
A Thermo Scientific Vanquish UPLC paired with a Q-Exactive tandem orbitrap mass spectrometer, operating in positive polarity mode with a nominal resolution of 70,000, was used to identify unknown compounds and to quantify caffeine in the residue sample. An ACE Excel 2 SuperC18 UPLC column (100 × 2.10 mm, 2 µm) was used to achieve separation of the compounds in the residue using a mobile phase consisting of: ultrapure water, methanol ( ≥ 99.8% purity, Honeywell), acetonitrile (≥99.9% purity, JT Baker), and 1 M acetic acid. All mobile phase components also contained 0.1% formic acid. The LC flow rate was 350 µL min–1, and the gradient used for the compound identification runs can be found in Supplementary Table 3. The MS system was run in full MS mode (over a m/z range of 150–1000) for compound identification. Identification of caffeine was confirmed by investigating the TIC peak located at the same retention time as that of a caffeine standard solution and matching the highest intensity peak in the mass spectrum at that retention time to that of protonated caffeine to less than 2 ppm of mass error. Further, there was agreement between the observed isotope pattern and a simulation of the expected pattern for caffeine generated using the Freestyle data analysis software package (Thermo Scientific), Supplementary Fig. 9.
Quantification of caffeine was performed using targeted selected ion monitoring (t-SIM) mode centered at m/z of 195.0876 with an isolation width of 1.0. The unknown, and each standard, was measured in triplicate, and the data processed using an automated peak-finding and peak-fitting algorithm available in the Freestyle data analysis application. Each sample was injected using the UPLC, and a short mobile phase gradient was used with a total run time of 8 min per injection. The details of this gradient can be found in Supplementary Table 4.
Roasting
A green coffee, El Tambo from Cauca, Colombia, was sourced from Tailored Coffee Roasters, Eugene, OR. This coffee is representative of an average specialty coffee, and costs ≈10 $ kg–1. The coffee was roasted in 50 g sample batches using an IKAWA Pro50. Sample batches were roasted using a simple roast profile, and the associated profiles are presented in Fig. 3a. The initial roast profile (roast 1a) went for 6 min 17 s and ended at 211.11 °C (412 °F). Additional roasts were each extended by an additional 30 s and 1.1 °C (2 °F) for a total of 8 min 17 s and an ending temperature of 215.56 °C (420 °F) for roast 1e. The roast development extension was doubled to 60 s and 2.2 °C (4 °F) between roasts 1e and 1f to ensure a sufficiently dark sample was included. Roast colors are presented on the Agtron Gourmet scale, where Agtron values range from 0 (black) to 150 (green, unroasted). The values were measured using the Coffee Dipper spectrophotometer in whole bean or ground mode, as necessary. The data is collated in Supplementary Table 4.
Sourced coffee samples
Coffee samples used in Fig. 4 were sourced from Colonna Coffee, Bath, UK. Those coffees were roasted to the same specifications with terminal whole bean colors of 98.9, 93.9, 92.8, and 93.6 Agtron units mapping to samples 1, 2, 3, and 4, respectively. Sample 1 was rejected on their cupping table, while the other three samples were accepted.
Computational models
All calculations were performed using the Vienna ab initio Simulation Package (VASP 5.4.4)68,69, using a plane wave basis set and projector augmented-wave pseudopotentials70. Bare surfaces of Pt(100), Pt(110), and Pt(111) as well as gas-phase molecules of caffeine and 5-CQA were geometrically optimized with density functional theory using a cutoff energy of 400 eV, the PBEsol functional71, and a k-point spacing of 0.03 2π Å–1 with a 20 Å vacuum layer. Interlayer van der Waals dispersion interactions were modeled using the DFT+D3 formalism with Becke-Johnson damping.72,73, The unit cells of Pt(hkl) contained 4 discrete layers of Pt atoms, and the ionic coordinates of the bottom 2 layers were frozen as bulk Pt, while the top 2 layers were allowed to relax. DFT optimizations of caffeine/Pt(hkl) and chlorogenic acid/Pt(hkl) were initialized from minimum energy configurations derived from molecular dynamics simulations and used a single k-point at Gamma. For caffeine/Pt(hkl), 4 layers of Pt were used with the bottom 2 layers frozen, and for chlorogenic acid/Pt(hkl), 2 layers of Pt were used with the bottom layer frozen. Molecular dynamics simulations were performed with a 300 eV cutoff energy using the Nosé-Hoover thermostat74,75, with 1 fs time steps beginning at 300 K and approaching 1 K, based on prior research on glycerol adsorption at Pt surfaces76. For both DFT and MD, the convergence criteria were 10 meV Å–1 for atomic forces and 1 × 10−6 eV for total energy.
Reporting summary
Further information on research design is available in the Nature Portfolio Reporting Summary linked to this article.
Data availability
The data that support the findings of this study are available from FigShare77 and from the corresponding author upon request. Source data are provided with this paper.
References
Sunarharum, W. B., Williams, D. J. & Smyth, H. E. “Complexity of coffee flavor: a compositional and sensory perspective. Food Res. Int. 62, 315–325 (2014).
Frost, S. C., Ristenpart, W. D. & Guinard, J. X. Effects of brew strength, brew yield, and roast on the sensory quality of drip brewed coffee. J. Food Sci. 85, 2530–2543 (2020).
Hu, G. et al. Effect of roasting degree of coffee beans on sensory evaluation: Research from the perspective of major chemical ingredients. Food Chem. 331, 127329 (2020).
Bhumiratana, N., Adhikari, K. & Chambers, E. “Evolution of sensory aroma attributes from coffee beans to brewed coffee. LWT Food Sci. Technol. 44, 2185–2192 (2011).
de Carvalho Pires, F. et al. “Feasibility of using colorimetric devices for whole and ground coffee roasting degrees prediction. J. Sci. Food Agric. 104, 5435–5441 (2024).
Lee, M. J., Kim, S. E., Kim, J. H., Lee, S. W. & Yeum, D. M. “A study of coffee bean characteristics and coffee flavors in relation to roasting. J. Korean Soc. Food Sci. Nutri. 42, 255–261 (2013).
de, C. et al. Near-infrared spectroscopy and multivariate calibration as an alternative to the Agtron to predict roasting degrees in coffee beans and ground coffee. Food Chem 365, 130471 (2021).
Miller, W. J. W. et al. “Using a Handheld refractometer in remote environments to measure the refractive indices of a variety of solutions: Sucrose, coffee, methanol/water, and 2-propanol/water. J. Chem. Educ. 98, 2730–2734 (2021).
Angeloni, G. et al. Characterization and comparison of cold brew and cold drip coffee extraction methods. J. Sci. Food. Agric. 99, 391–399 (2019).
Batali, M. E., Ristenpart, W. D. & Guinard, J. X. Brew temperature, at fixed brew strength and extraction, has little impact on the sensory profile of drip brew coffee. Sci. Rep. 10, 16450 (2020).
Cameron, M. I. et al. “Systematically improving espresso: insights from mathematical modeling and experiment. Matter 2, 631–648 (2020).
Lingle, T. R. The Coffee Brewing Handbook A Systematic Guide to Coffee Preparation (Specialty Coffee Association, 1996).
Guinard, J. X. et al. A new Coffee Brewing Control Chart relating sensory properties and consumer liking to brew strength, extraction yield, and brew ratio. J. Food Sci. 88, 2168–2177 (2023).
Spiro, M. & Chong, Y. Y. “The kinetics and mechanism of caffeine infusion from coffee: The temperature variation of the hindrance factor. J. Sci. Food. Agric. 74, 416–420 (1997).
Gloess, A. N., Yeretzian, C., Knochenmuss, R. & Groessl, M. On-line analysis of coffee roasting with ion mobility spectrometry–mass spectrometry (IMS–MS). Int. J. Mass. Spectrom. 424, 49–57 (2018).
Cordoba, N., Fernandez-Alduenda, M., Moreno, F. L. & Ruiz, Y. “Coffee extraction: A review of parameters and their influence on the physicochemical characteristics and flavour of coffee brews. Trends Food Sci. Technol. 96, 45–60 (2020).
Mestdagh, F., Davidek, T., Chaumonteuil, M., Folmer, B. & Blank, I. “The kinetics of coffee aroma extraction. Food Res. Int. 63, 271–274 (2014).
Castañeda-Rodríguez, R., Mulík, S. & Ozuna, C. “Brewing temperature and particle size affect extraction kinetics of cold brew coffee in terms of its physicochemical, bioactive, and antioxidant properties. J. Culin. Sci. Technol. 20, 366–387 (2022).
Spiro, M. & Hunter, J. E. “The kinetics and mechanism of caffeine infusion from coffee: The effect of roasting. J. Sci. Food Agric. 36, 871–876 (1985).
Munteanu, I. G. & Apetrei, C. A review on electrochemical sensors and biosensors used in chlorogenic acid electroanalysis. Int. J. Mol. Sci. 22, 13138 (2021).
Švorc, Ľ “Determination of caffeine: a comprehensive review on electrochemical methods. Int. J. Electrochem. Sci. 8, 5755–5773 (2013).
Redivo, L. et al. Ĺ. “Bare carbon electrodes as simple and efficient sensors for the quantification of caffeine in commercial beverages. R. Soc. Open Sci. 5, 172146 (2018).
Tomac, I. & Šeruga, M. “Electrochemical properties of chlorogenic acids and determination of their content in coffee using differential pulse voltammetry. Int. J. Electrochem. Sci. 11, 2854–2876 (2016).
Ziyatdinova, G., Aytuganova, I., Nizamova, A. & Budnikov, H. “Differential pulse voltammetric assay of coffee antioxidant capacity with MWNT-Modified electrode. Food Anal. Methods 6, 1629–1638 (2013).
Ly, S. Y. et al. “Determination of caffeine using a simple graphite pencil electrode with square-wave anodic stripping voltammetry. Microchimica Acta 146, 207–213 (2004).
D’Amelio, N., Fontanive, L., Uggeri, F., Suggi-Liverani, F. & Navarini, L. NMR reinvestigation of the caffeine–chlorogenate complex in aqueous solution and in coffee brews. Food Biophys. 4, 321–330 (2009).
Kilmartin, P. A. & Hsu, C. F. “Characterisation of polyphenols in green, oolong, and black teas, and in coffee, using cyclic voltammetry. Food Chem. 82, 501–512 (2003).
Alpar, N., Yardım, Y. & Şentürk, Z. “Selective and simultaneous determination of total chlorogenic acids, vanillin and caffeine in foods and beverages by adsorptive stripping voltammetry using a cathodically pretreated boron-doped diamond electrode. Sens. Actuators B Chem. 257, 398–408 (2018).
Švorc, L., Tomčík, P., Svítková, J., Rievaj, M. & Bustin, D. Voltammetric determination of caffeine in beverage samples on bare boron-doped diamond electrode. Food Chem. 135, 1198–1204 (2012).
Yardim, Y., Keskin, E. & Şentürk, Z. Voltammetric determination of mixtures of caffeine and chlorogenic acid in beverage samples using a boron-doped diamond electrode. Talanta 116, 1010–1017 (2013).
Sarakhman, O. & Švorc, Ľ A review on recent advances in the applications of boron-doped diamond electrochemical sensors in food analysis. Crit. Rev. Anal. Chem. 52, 791–813 (2022).
Sebokolodi, T. I., Sipuka, D. S., Tsekeli, T. R., Nkosi, D. & Arotiba, O. A. “An electrochemical sensor for caffeine at a carbon nanofiber modified glassy carbon electrode. J. Food Meas. Charact. 16, 2536–2544 (2022).
Amare, M. & Aklog, S. Electrochemical determination of caffeine content in ethiopian coffee samples using lignin modified glassy carbon electrode. J. Anal. Methods Chem. 2017, 3979068 (2017)
Tajeu, K. Y., Dongmo, L. M. & Tonle, I. K. “Fullerene/MWCNT/nafion modified glassy carbon electrode for the electrochemical determination of caffeine. Am. J. Analyt. Chem. 11, 114–127 (2020).
Amare, M. & Admassie, S. Polymer modified glassy carbon electrode for the electrochemical determination of caffeine in coffee. Talanta 93, 122–128 (2012).
Gupta, V. K., Jain, A. K. & Shoora, S. K. “Multiwall carbon nanotube modified glassy carbon electrode as voltammetric sensor for the simultaneous determination of ascorbic acid and caffeine. Electrochim. Acta 93, 248–253 (2013).
Navarini, L. & Rivetti, D. “Water quality for Espresso coffee. Food Chem 122, 424–428 (2010).
Rao, N. Z. & Fuller, M. Acidity and antioxidant activity of cold brew coffee. Sci. Rep. 8, 16030 (2018).
Rao, N. Z., Fuller, M. & Grim, M. D. Physiochemical characteristics of hot and cold brew coffee chemistry: the effects of roast level and brewing temperature on compound extraction. Foods 9, 902 (2020).
Conway, B. E., Angerstein-Kozlowska, H., Sharp, W. B. A. & Criddle, E. E. “Ultrapurification of water for electrochemical and surface chemical work by catalytic pyrodistillation. Anal. Chem. 45, 1331–1336 (1973).
Kondo, T., Masuda, T., Aoki, N. & Uosaki, K. “Potential-dependent structures and potential-induced structure changes at Pt(111) single-crystal electrode/sulfuric and perchloric acid interfaces in the potential region between hydrogen underpotential deposition and surface oxide formation by in situ surface X-ray scattering. J. Phys. Chem. C 120, 16118–16131 (2016).
Herrero, E., Buller, L. J. & Abruña, H. D. Underpotential deposition at single crystal surfaces of Au, Pt, Ag and other materials. Chem. Rev. 101, 1897–1930 (2001).
Ishikawa, Y., Mateo, J. J., Tryk, D. A. & Cabrera, C. R. “Direct molecular dynamics and density-functional theoretical study of the electrochemical hydrogen oxidation reaction and underpotential deposition of H on Pt(111). J. Electroanal. Chem. 607, 37–46 (2007).
Łosiewicz, B., Jurczakowski, R. & Lasia, A. “Kinetics of hydrogen underpotential deposition at polycrystalline platinum in acidic solutions. Electrochim. Acta 80, 292–301 (2012).
Jerkiewicz, G. “Electrochemical hydrogen adsorption and absorption. Part 1: under-potential deposition of hydrogen. Electrocatal 1, 179–199 (2010).
Saba, P., Brown, W. A. & Omanovic, S. “Interactive behavior of caffeine at a platinum electrode surface. Mater. Chem. Phys. 100, 285–291 (2006).
Hoshi, N., Nakamura, M., Kubo, R. & Suzuki, R. Enhanced oxygen reduction reaction on caffeine-modified platinum single-crystal electrodes. Commun. Chem. 7, 23 (2024).
Deblois, M., Lessard, J. & Jerkiewicz, G. “Influence of benzene on the HUPD and anion adsorption on Pt(110), Pt(100) and Pt(111) electrodes in aqueous H2SO4. Electrochim. Acta 50, 3517–3523 (2005).
Martí-Rujas, J. & Kawano, M. Kinetic products in coordination networks: Ab initio X-ray powder diffraction analysis. Acc. Chem. Res. 46, 493–505 (2013).
Hečimović, I., Belščak-Cvitanović, A., Horžić, D. & Komes, D. Comparative study of polyphenols and caffeine in different coffee varieties affected by the degree of roasting. Food Chem. 129, 991–1000 (2011).
Belay, A., Ture, K., Redi, M. & Asfaw, A. “Measurement of caffeine in coffee beans with UV/vis spectrometer. Food Chem. 108, 310–315 (2008).
Bell, L. N., Wetzel, C. R. & Grand, A. N. “Caffeine content in coffee as influenced by grinding and brewing techniques. Food Res. Int. 29, 785–789 (1996).
Kamiyama, M., Moon, J.-K., Jang, H. W. & Shibamoto, T. Role of degradation products of chlorogenic acid in the antioxidant activity of roasted coffee. J. Agric. Food. Chem. 63, 1996–2005 (2015).
Moon, J.-K., Yoo, H. S. & Shibamoto, T. Role of roasting conditions in the level of chlorogenic acid content in coffee beans: correlation with coffee acidity. J. Agric. Food. Chem. 57, 5365–5369 (2009).
Akinola, J., Barth, I., Goldsmith, B. & Singh, N. Adsorption energies of oxygenated aromatics and organics on rhodium and platinum in aqueous phase. ACS Catal 10, 4929–4941 (2020).
Shetty, M., Ardagh, M. A., Pang, Y., Abdelrahman, O. A. & Dauenhauer, P. J. Electric-field-assisted modulation of surface thermochemistry. ACS Catal. 10, 12867–12880 (2020).
Lindsey, Z., Williams, J. R., Burgess, J. S., Moore, N. T. & Splichal, P. M. Caffeine content in filter coffee brews as a function of degree of roast and extraction yield. Sci. Rep. 14, 29126 (2024).
Wang, X. & Lim, L. T. “Effect of roasting conditions on carbon dioxide degassing behavior in coffee. Food Res. Int. 61, 144–151 (2014).
Di Donfrancesco, B., Gutierrez Guzman, N. & Chambers, E. “Comparison of results from cupping and descriptive sensory analysis of colombian brewed coffee. J. Sens. Stud. 29, 301–311 (2014).
Tarigan, E. B., Wardiana, E., Hilmi, Y. S. & Komarudin, N. A. The changes in chemical properties of coffee during roasting: a review. IOP Conf. Ser. Earth Environ. Sci. 974, 012115 (2022).
Rojas-González, A. et al. Coffee chlorogenic acids incorporation for bioactivity enhancement of foods: a review. Molecules 27, 3400 (2022).
Batali, M. E., Cotter, A. R., Frost, S. C., Ristenpart, W. D. & Guinard, J.-X. “Titratable acidity, perceived sourness, and liking of acidity in drip brewed coffee. ACS Food Sci. Technol. 1, 559–569 (2021).
Poole, R. L. & Tordoff, M. G. The taste of caffeine. J. Caffeine Res. 7, 39–52 (2017).
Linne, B. M., Tello, E., Simons, C. T. & Peterson, D. G. Characterization of the impact of chlorogenic acids on tactile perception in coffee through an inverse effect on mouthcoating sensation. Food Res. Int. 172, 113167 (2023).
Gao, C., Tello, E. & Peterson, D. G. Identification of coffee compounds that suppress bitterness of brew. Food Chem 350, 129225 (2021).
Hendon, C. H. “Chemistry and coffee. Matter 2, 514–518 (2020).
Daubinger, P., Kieninger, J., Unmüssig, T. & Urban, G. A. Electrochemical characteristics of nanostructured platinum electrodes - a cyclic voltammetry study. Phys. Chem. Chem. Phys. 16, 8392–8399 (2014).
Kresse, G. & Furthmüller, J. Efficient iterative schemes for ab initio total-energy calculations using a plane-wave basis set. Phys. Rev. B 54, 11169–11186 (1996).
Kresse, G. & Furthmüller, J. Efficiency of Ab-initio total energy calculations for metals and semiconductors using a plane-wave basis set. Comp. Mat. Sci. 6, 15–50 (1996).
Kresse, G. & Joubert, D. From ultrasoft pseudopotentials to the projector augmented-wave method. Phys. Rev. B 59, 1758–1775 (1999).
Perdew, J. P. et al. Restoring the density-gradient expansion for exchange in solids and surfaces. Phys. Rev. Lett. 100, 136406 (2008).
Grimme, S., Antony, J., Ehrlich, S. & Krieg, H. A consistent and accurate ab initio parametrization of density functional dispersion correction (DFT-D) for the 94 elements H-Pu. J. Chem. Phys. 132, 154104 (2010).
Grimme, S., Ehrlich, S. & Goerigk, L. Effect of the damping function in dispersion corrected density functional theory. J. Comp. Chem. 32, 1456–1465 (2011).
Nosé, S. Constant temperature molecular dynamics methods. Prog. Theor. Phys. Suppl. 103, 1–46 (1991).
Hoover, W. G. Canonical dynamics: equilibrium phase-space distributions. Phys. Rev. A 31, 1695–1697 (1985).
Tereshchuk, P., Chaves, A. S. & Da Silva, J. L. F. Glycerol adsorption on platinum surfaces: a density functional theory investigation with van der Waals corrections. J. Phys. Chem. C 118, 15251–15259 (2014).
Hendon, C. H. “Direct electrochemical appraisal of black coffee quality using cyclic voltammetry” FigShare, https://doi.org/10.6084/m9.figshare.30811541 (2026).
Acknowledgements
This work was supported by the Coffee Science Foundation, underwritten by Nuova Simonelli. The authors are also grateful for the support from the National Science Foundation under grant no. 2237345 and the Camille and Henry Dreyfus Foundation. We are grateful to Dr. Jiawei Huang for his help performing QCM experiments. The authors also appreciate many helpful discussions with Dr. Andreas Stonas and Brian Sung. We are grateful to Maxwell Dashwood at Colonna Coffee for providing the four samples of roasted coffee. Other coffee used in this study inevitably came from in-house roasting but the methods were developed using coffees donated to the laboratory by Counter Culture Coffee, Onyx Coffee Lab, Sweetbloom, Moongoat, Passenger, Black and White, Tim Wendelboe, Monogram, Archetype, Farmers Union, Tailored, Reverie, Blueprint, ONA, Phil and Sebastian, Proud Mary, Keurig Dr. Pepper, Mostra, Coffea Circulor, Mother Tongue, Creature, and Intelligentsia.
Ethics declarations
Competing interests
C.H.H. and D.L.P. have a financial interest in Overpotential, a company commercializing electrochemically modified food. Apart from this, the authors declare no competing interests.
Peer review
Peer review information
Nature Communications thanks Zachary Lindsey, Hitoshi Muguruma, Kenshin Takemura, and the other, anonymous, reviewer for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Bumbaugh, R.E., Pennington, D.L., Wehn, L.C. et al. Direct electrochemical appraisal of black coffee quality using cyclic voltammetry. Nat Commun 17, 3618 (2026). https://doi.org/10.1038/s41467-026-71526-5
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41467-026-71526-5