Introduction
Coffee is a plant-based beverage made from processed coffee beans. Its flavour and composition vary based on the bean type, ripeness, processing, roasting, and brewing methods1. Key phytochemicals in coffee include alkaloids (like caffeine), (poly)phenols (such as phenolic acids), diterpenes, and melanoidins formed during roasting1,2. Moderate coffee consumption is associated with various health benefits, including reduced risks of type 2 diabetes, liver disease, cardiovascular diseases, and cancer3. In a large cross-sectional study of 468,629 individuals without clinical cardiovascular disease, light-to-moderate coffee consumption was linked to lower rates of all-cause mortality, cardiovascular mortality, and stroke incidence4. Furthermore, coffee intake is consistently associated with a reduced risk of Parkinson’s disease in a dose-dependent manner, across multiple human cohorts5,6,7. Meta-analyses have also found that coffee consumers face a lower risk of depression8,9, and one meta-analysis of cohort studies examining cognitive decline, showed that coffee consumption accounted for a 27% reduction in the incidence of Alzheimer’s disease10.
These systemic benefits are paralleled by evidence that coffee impacts the brain directly, shaping both neural activity and cognitive outcomes. Functional MRI studies show that habitual coffee drinkers exhibit altered functional connectivity in regions linked to sensory, motor, and emotional processing. These changes appear to depend on frequency of intake, suggesting that regular coffee consumption may influence brain function and emotional regulation11. Recent data show a positive correlation between cognitive performance, particularly memory and processing speed, in older adults12. Coffee consumption causes a temporary rise in cortisol levels, followed by normalization with habitual use, indicating physiological adaptation13. However, the effects of coffee on stress remain inconclusive, with studies showing mixed results14,15,16.
Coffee also affects the gastrointestinal tract. It increases stomach acidity and stimulates the release of hormones that aid digestion. Both caffeinated and decaffeinated coffee promote the contractility of ileal and colonic smooth muscle, helping prevent constipation17,18. It is becoming clear that coffee influences human health and physiology through both direct and indirect mechanisms. Direct effects arise from interactions between coffee compounds or their circulating metabolites and specific biological targets, while indirect effects involve changes to systems such as possibly via the gut microbiome.
A growing body of evidence from in vitro, animal, and human studies suggests that the microbes in the gut are also reactive to coffee, and that coffee has prebiotic-like effects on the gut microbiome19. This is primarily due to its fiber-like compounds and phenolics such as chlorogenic acids (CGAs) including caffeoylquinic, feruloylquinic, and coumaroylquinic acids. For example, melanoidins in coffee may increase serum short-chain fatty acid (SCFA) levels by promoting the growth of SCFA-producing bacteria20,21,22. Moreover, some studies suggest that coffee consumption promotes growth of Bacteroides and probiotic species belonging to the Bifidobacterium and Lactobacillus genera20,23,24,25,26,27,28. The bioavailability and metabolism of coffee (poly)phenols vary widely between individuals, a phenomenon influenced by the gut microbiome29,30. Coffee phenolics may also reduce neuroinflammation by activating antioxidant response factors in the brain31,32,33,34
A recent metagenomics study involving over 1000 participants examined the relationship between more than 150 dietary components and the composition of the gut microbiome. This study identified coffee consumption as the most strongly correlated food item in a dose-dependent manner; subsequently validating it in a second cohort35. It is possible that coffee’s influence on cognition is mediated by its interaction with the gut microbiome. For instance, coffee consumption is linked to higher levels of beneficial butyrate-producing gut bacteria such as Lawsonibacter asaccharolyticus36. Indeed, there is a growing appreciation of the importance of the impact of food and beverages on the bidirectional signalling pathways that make up the microbiota-gut-brain axis37.
Although epidemiological and mechanistic studies strongly associate coffee intake with improved health outcomes, key gaps remain. First, the temporal dynamics of coffee consumption, withdrawal, and reintroduction remain poorly understood. Second, the role of individual variability in metabolizing coffee phenolics, shaped by the gut microbiome, has not been systematically examined. Finally, the contribution of the microbiome as a mediator between coffee intake and brain function has yet to be established.
In this study, we aim to explore the temporal effects of coffee consumption, withdrawal, and reintroduction on cognition, mood, and behavior through self-reported questionnaires in the context of the microbiota-gut-brain axis. Thus, concurrent with behavioural analysis, we simultaneously profile the gut microbiota of healthy, adult, moderate coffee drinkers compared with non-coffee drinkers, using shotgun metagenomics, and targeted/untargeted metabolomics. In addition, different pathways of the microbiota-gut-brain axis such as stress, inflammation, and microbial-derived metabolites are explored38. We aim to interrogate the overlap between coffee’s influence on the microbiome and cognition through several mechanisms. Through (poly)phenols39, by the positive benefit of increased butyrate-producing bacteria36 or alteration of the gut-brain axis with coffee altering neuroactive compounds such as SCFAs or γ-aminobutyric acid17. Furthermore, we probe the inter-individual variability in the metabolism of coffee phenolics and how the microbiome may influence this. While this study considers the role of caffeine, coffee, as a complex mixture, contains many other compounds that may influence the gut.
We hypothesize that coffee consumption will have distinct effects on the gut microbiome, increasing microbial diversity and beneficial mediators of gut health, some of which may depend on caffeine, while others may not. These changes may contribute to improved brain function via enhanced stress resilience and cognitive performance, thereby linking coffee consumption to the microbiota–gut–brain axis via a unique interaction between coffee, the microbiome, and cognition.
Results
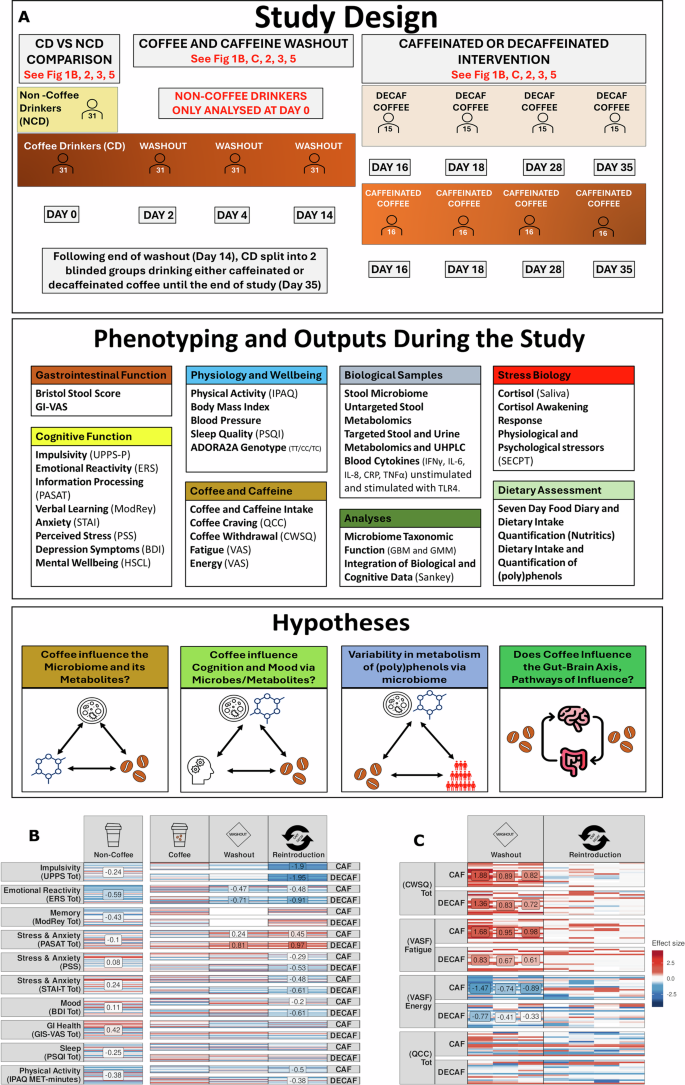
This prospective study had three phases each evaluating cognition, stress, physical health, mood, immune function, gut microbiome, dietary intake, and metabolite composition (see Fig. 1 for design). Firstly, we compared non-coffee drinkers (NCD, n = 31) with coffee drinkers (CD, n = 31), then the CD group abstained from coffee for 14 days. Following this washout period, participants reintroduced either decaffeinated (n = 15) or caffeinated coffee (n = 16) for 21 days (Fig. 1A). Participants in the NCD group did not participate in any subsequent phases of the study. Overall, more females participated, and most participants were born per vaginum (Table 1). At baseline, CD participants consumed more caffeine daily than NCDs, and NCDs gave up more caffeine before the baseline visit (Table 1, Supplementary Data 1), though the level given up was substantially lower than what would be considered addictive. Alcohol consumption, education years, Childhood Trauma Questionnaire (CTQ) sub-scores, and predicted IQ (measured using the National Adult Reading Test (NART)) did not differ between groups (Table 1). When comparing these metrics between caffeinated and decaffeinated coffee drinkers during the intervention phase of the study, daily caffeine intake, the amount of caffeine abstained from, alcohol consumption, education years, CTQ sub-scores, and predicted IQ did not differ between these groups (Supplementary Data 15). All participants were genotyped for caffeine sensitivity-related SNPs rs2298383 and rs5751876 of the ADORA2A gene. NCDs had a higher percentage of the C/C haplotype in rs2298383, while CDs had more C/T. For rs5751876, NCDs had more T/T, and CDs had more C/T (Table 1, Supplementary Fig. 1). In previous research, the T/T haplotype has been associated with higher coffee intake40. This discrepancy may suggest that contextual, environmental, or sample-specific factors moderated the relationship between genotype and caffeine consumption in this population.
A Experimental overview, NCD and CD drinkers at baseline (V2), CD drinkers following 2 weeks without coffee (V3) and 3 weeks following reintroduction of caffeinated or decaffeinated coffee. Sample type, questionnaire type are indicated in each panel along with day of study. B Heatmap of questionnaire results relative to baseline CD. Horizontal bars within the same cell represent values of individual participants. For the coffee drinker subset, which features repeated measurements, measurements from the same participant are aligned on the y-axis. Red colour indicates positive effect size while blue reflects negative effect size, with intensity of red or blue corresponding to increased or decreased effect sizes respectively and white reflecting zero effect. Panels containing a text box with numbers reflects those comparisons with a Cohen’s d effect size of > 0.5 reflecting medium sized effects. Column one represents questionnaire completed, column two = NCD, column three = CD, column four washout period for CD and column five reintroduction of caffeinated or decaffeinated coffee. C Heatmap of questionnaire responses to craving and fatigue symptoms following caffeine abstinence and reintroduction of caffeinated or decaffeinated coffee for CD. Column one represents the questionnaire completed, column two = washout period for CD and column three reintroduction of caffeinated or decaffeinated coffee. Comparisons are NCD v CD, washout v CD baseline and CAF or DECAF v CD baseline values. n = 31 at baseline for CD and NCD, n = 31 during washout period, n = 15 during reintroduction of decaffeinated coffee and N = 16 during reintroduction of caffeinated coffee. Abbreviations: UPPS-P, urgency premeditation perseverance sensation seeking positive urgency impulsive behaviour scale; ERS, emotional and reactivity scale, PASAT, paced auditory serial addition test; ModRey, modified auditory verbal learning test, STAI (Trait), state trait anxiety inventory; PSS, perceived stress scale; BDI, Beck’s depression inventory; GIS-VAS, gastrointestinal symptoms visual analogue scale; HSCL GSI, Hopkins symptom checklist general severity index; PSQI, Pittsburgh sleep quality index; IPAQ, international physical activity questionnaire; QCC, questionnaire of caffeine cravings, CWSQ, caffeine withdrawal symptom questionnaire, V visit.
Participants’ food intake was monitored for 7 days with a weighted food diary before each study visit (See Supplementary File). No differences in dietary intake were observed when comparing CD participants with NCD participants (Supplementary Data 2a). After 2 weeks of caffeine abstinence, dietary intake remained largely unchanged within CD, though the caffeinated group reported slightly lower magnesium intake, which remained unchanged until the end of the study, while the decaffeinated group had a slight increase in selenium intake from their baseline to their endpoint visit (Supplementary Data 2b–e). No other differences in dietary intakes of macro-, micronutrients, and food groups were observed.
With no significant dietary differences between CD and NCD at baseline, we assessed general well-being using self-reported questionnaires. At baseline, comparing CD with NCD, there were no differences in body mass index (BMI), blood pressure, stress, anxiety, depression, gastrointestinal symptoms, sleep quality, or physical activity (Supplementary Data 3). After the caffeine washout period, these measures also remained unchanged (Supplementary Data 4). During the intervention phase, CD participants showed a significant decrease in systolic and diastolic blood pressure after 2 weeks of coffee abstinence (Supplementary Data 5). BMI remained unchanged in both caffeinated and decaffeinated groups after the washout and coffee intervention while those consuming caffeinated coffee had a decrease in systolic blood pressure compared with the decaffeinated group (Supplementary Data 6).
Behavioural and cognitive modulation by coffee consumption and withdrawal in habitual and non-habitual drinkers
Comparing the cognitive status of NCD and CD participants using self-reported questionnaires, CDs scored significantly higher on the impulsivity behavior scale (UPPS-P) and its “Sensation Seeking” sub-score, indicating a greater motivation for novelty (Fig. 1B, Supplementary Data 3). CDs also had higher scores on the emotion reactivity scale (ERS) compared to NCDs (Fig. 1B, Supplementary Data 3). After habitual coffee drinkers abstained from drinking coffee, both UPPS-P total and “Negative Urgency” scores, as well as ERS scores for sensitivity, intensity, persistency, and the total ERS score, were significantly lower than baseline (Fig. 1B, Supplementary Data 4). Also, after the coffee washout period, participants showed improved cognitive performance on the PASAT test, possibly due to task repetition (Supplementary Data 4).
To examine the potential difference in these self-reported responses between caffeinated and decaffeinated coffee recipients during the intervention phase of the study, we compared questionnaire responses at the beginning with responses at the end of the intervention. The caffeinated group had reduced anxiety (STAI, Trait) and psychological distress (HSCL), while both the caffeinated and the decaffeinated group improved in perceived stress, BDI and UPPS-P (Supplementary Data 5). Recipients of caffeinated coffee also had increased scores on the PASAT (Supplementary Data 5) which suggests that improvements in cognitive performance post-washout were not solely due to task repetition. Comparing self-reported questionnaire responses between the start of study with the period post-intervention, both groups showed decreases in UPPS-P and ERS total scores and sensitivity from the beginning to the end of the study. ERS intensity decreased only in decaffeinated coffee drinkers (Supplementary Data 6). Post-intervention, total impulsivity and emotional reactivity scores decreased compared to scores at the start of the intervention (Supplementary Data 6). In caffeinated coffee drinkers, perseverance and negative urgency decreased, while decaffeinated drinkers showed reductions in positive urgency. The STAI-Trait total score decreased in caffeinated drinkers, while HSCL, sleep quality (PSQI), and physical activity (IPAQ) scores improved in decaffeinated drinkers (Supplementary Data 6) suggesting that caffeinated and decaffeinated coffee may have distinct psychological and physiological benefits, potentially influencing recommendations for their consumption based on individual health goals.
The ModRey episodic memory test was used to assess declarative memory performance in participants. At the baseline visit, performance was similar between CD and NCD with CD and NCD improving in delayed recall at the baseline (Supplementary Data 7). When comparing ModRey scores between the start of the study, at baseline, and the end of the study, post-intervention significant improvement was seen only in participants receiving decaffeinated coffee during the intervention phase of the study (Supplementary Data 8). The decaffeinated group demonstrated significant improvements in several ModRey test components, whereas the caffeinated group did not. These findings may be attributable to the beneficial impact of improved sleep quality and increased physical activity, both of which are well-documented to support episodic memory performance41,42.
At baseline, there was no difference between CD and NCD when assessing emotional processing using the emotion recognition task (ERT). Subsequently following the intervention, in both caffeinated and decaffeinated participants, there were no significant differences in ERT performance post-intervention compared to their scores at baseline (Supplementary Data 8). At baseline, comparing CD with NCD there was no significant difference between learning and forgetting information using the PAL task (Supplementary Data 7). However, following the intervention the decaffeinated group showed a significant decrease in PAL errors, unlike the caffeinated group, suggesting that non-caffeine components of coffee may improve learning and memory (Supplementary Data 8) though, there is also a chance of learning effect.
To assess the effect of caffeine withdrawal on CD during the 2-week coffee withdrawal, self-report questionnaires were administered at day 2, day 4 and on the day the intervention started (day 14) (Fig1A). Craving scores remained stable across this period (Fig. 1C and Supplementary Fig. S8). Across the same period, symptoms of withdrawal were elevated at the start of the withdrawal phase and dropped significantly at day 4 and visit 3 (intervention). All measures, including drowsiness and headaches significantly reduced (Supplementary Fig. S9). Using self-reported VAS measures of fatigue and energy, we saw reduced fatigue and increased energy in CD participants across the withdrawal period (Supplementary Fig. S9).
Using the same self-reported questionnaire of caffeine withdrawal symptoms at 4 timepoints across the intervention phase we saw that overall, there was little change in withdrawal symptoms other than increased drowsiness and fatigue in decaffeinated coffee drinkers (Supplementary Fig. S10a) We also saw an increase in “decreased sociability, motivation to work” in decaffeinated participants at the start of the study, while in the caffeinated participants there was a significant temporal increase at T4 and a significant decrease in “decreased sociability, motivation to work” (Supplementary Fig S10d). These findings suggest that consumption of coffee containing caffeine improves mood and reduces withdrawal symptoms (Fig. 1C and Supplementary Fig. 8-11).
Overall, we see that impulsivity and emotional reactivity are influenced by coffee; CD participants exhibit higher levels of both compared to NCD participants, and abstinence from coffee reduces these measures. The reintroduction of caffeinated or decaffeinated coffee significantly reduces impulsivity, while decaffeinated coffee alone reduces emotional reactivity. Caffeinated coffee improved anxiety and some aspects of cognitive performance, while decaffeinated coffee led to gains in memory, sleep quality, and physical activity, suggesting distinct benefits based on coffee type. Overall, coffee intake and withdrawal both modulated self-reported behavior and physiological responses, with notable improvements in mood, cognition, and memory.
Mood benefits of coffee occur without changes in cortisol physiology
When considering stress and cortisol physiology, the Socially Evaluated Cold Pressor Test (SECPT), a stress-inducing task combining physical discomfort and social evaluation, showed a nonsignificant trend of increased self-reported anxiety (ΔSTAI - State) in NCDs compared to CDs, indicating higher anxiety in NCDs before the test (Supplementary Data 9). During the intervention, participants receiving caffeinated and decaffeinated coffee both showed lower perceived stress, depression, and impulsivity scores, indicating coffee consumption improves mood regardless of caffeine content (Supplementary Data 10). There was no difference in salivary cortisol levels between CD and NCD during the SECPT performed at the beginning of the study (Supplementary Fig. S3a, b). Salivary cortisol levels during the SECPT did not differ from baseline levels in participants, regardless of whether they consumed caffeinated or decaffeinated coffee during the intervention (Supplementary Fig. S7). During SECPT, both groups had minimal changes in self-reported questionnaires examining stress, mood, and anxiety. The caffeinated group showed increased “self-concept” and “secondary appraisal” on the Primary Appraisal Secondary Appraisal (PASA) questionnaire, indicating better coping, while the decaffeinated group reported higher Positive Affect Schedule scores, indicating elevated positive feelings (Supplementary Data 9).
Assessing baseline stress reactivity during the early morning cortisol surge, each participant performed the cortisol awakening response (CAR). Cortisol levels were similar between CD and NCD groups at baseline (Supplementary Fig. S3a, b). Similarly, no changes in CAR were observed after the coffee washout (Supplementary Fig.S4), indicating caffeine withdrawal did not affect cortisol levels in CD. During the intervention phase of the study, the caffeinated group had lower salivary cortisol upon awakening post-intervention (Supplementary Fig. 5a, c). The CAR of the decaffeinated group remained unchanged (Supplementary Fig. S5b, d) while basal stress levels and resilience during SECPT showed no differences in salivary cortisol for either group (Supplementary Data S9).
Coffee consumption alters immune responses without affecting gastrointestinal function
Examining cytokine secretion in peripheral blood, at baseline, CRP levels were lower in CD, while IL-10 levels increased compared to NCD (Supplementary Fig 13, Supplementary Data S,11). Coffee abstinence also led to changes in cytokine levels with increases in CRP and TNFα levels, suggesting a protective role of coffee or caffeine against inflammation (Supplementary Fig 13, Supplementary Data S,12). At the end of the intervention phase, the caffeinated group exhibited lower IL-10 and IL-6 levels (Supplementary Fig 13, Supplementary Data S,13) compared to pre-intervention. At the end of the reintroduction phase of the study, decaffeinated coffee consumption increased CRP and TNFα levels, while caffeine intake decreased IL-10 and IL-6 levels (Supplementary Fig 13 and Supplementary Data 11–14).
Following ex vivo stimulation of whole blood with LPS, NCD participants displayed a greater IL-6 increase upon TLR4 stimulation than CDs at baseline. Interestingly, both caffeinated and decaffeinated groups showed decreased IL-6 levels upon LPS stimulation post-intervention, suggesting other coffee components may influence immune responsiveness. Following two weeks without coffee, there was a significant increase in IL-6 secretion compared with secretion at baseline. When comparing stimulated cytokine secretion at the start and at the end of the study, there was significant reduction in TNFα secretion in participants drinking decaffeinated coffee compared with cytokine levels at baseline (Supplementary Fig. S14 and Supplementary Data 14).
Assessment of self-reported GI function using the Bristol Stool Chart revealed modest differences in stool consistency between CD and NCD, with a decrease in stool score in CD at baseline. After caffeine abstinence stool score increased in CD participants compared to their baseline score (Supplementary Fig. 2 and Supplementary Fig. 12). There were no significant changes in stool score at any timepoint throughout the intervention phase of the study.
Coffee consumption shapes fecal metabolite profiles through caffeine and microbial pathways
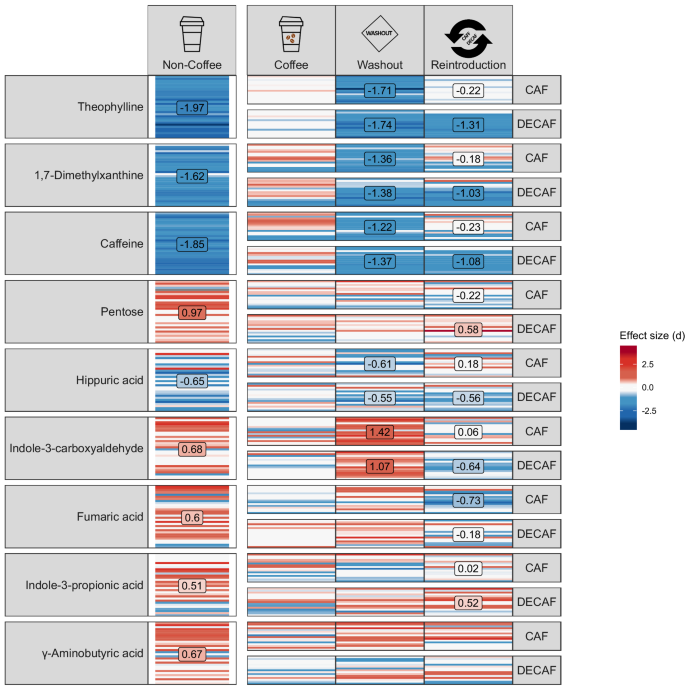
When we compared untargeted faecal metabolite levels between NCD and CD participants, we inevitably saw changes in the level of caffeine in the faeces of CD participants (Fig. 2). In addition, we observed significantly higher concentrations in theophylline, 1,7-dimethylxanthine, and hippuric acid for CD in comparison to NCD. Theophylline is a coffee alkaloid and 1,7-dimethylxanthine is a caffeine metabolite, while hippuric acid is a phenolic metabolite derived from coffee and other (poly)phenol dietary sources or the endogenous metabolism of aromatic amino acids and catecholamines43. In CD participants, indoles such as indole-3-propionic acid (IPA) and indole-3-carboxyaldehyde (ICA) and the neurotransmitter γ-aminobutyric acid (GABA) were significantly decreased. Indoles can be the product of microbial metabolism, suggesting complex metabolic responses to coffee consumption. In CD participants, following a 2-week period of coffee abstinence, level of caffeine, theophylline, 1,7-dimethylxanthine, and hippuric acid were significantly reduced, while ICA was significantly increased, suggesting, based on levels in NCD also, that ICA is a metabolite sensitive to the absence of caffeine. Following the reintroduction of either caffeinated or decaffeinated coffee, there were differing responses in metabolite concentration in faeces. Notably, caffeine, theophylline, hippuric acid and 1,7-dimethylxanthine did not return to the levels of baseline measurement, while pentose, hippuric acid and fumaric acid all had different concentrations depending on the presence of caffeine (Fig. 2).
Heatmap of untargeted faecal metabolomics from relative to baseline coffee drinkers (CD). Horizontal bars within the same cell represent values of individual participants. For the coffee drinker subset, which features repeated measurements, measurements from the same participant are aligned on the y-axis. Red colour indicates positive effect size while blue reflects negative effect size, with intensity of red or blue corresponding to increased or decreased effect sizes respectively and white reflecting zero effect. Panels containing a text box with numbers reflects those comparisons with a Cohen’s d effect size of > 0. 5 reflecting medium sized effects. Comparisons are NCD v CD, washout v CD baseline and CAF or DECAF v CD baseline values. Column one =metabolite, column two = NCD, column three = CD, column four washout period for CD and column five reintroduction of caffeinated or decaffeinated coffee. n = 31 at baseline for CD and NCD, n = 31 during washout period, n = 15 during reintroduction of decaffeinated coffee and n = 16 during reintroduction of caffeinated coffee.
Coffee consumption drives characteristic urinary metabolite signatures reflecting caffeine and polyphenol metabolism
The metabolism of caffeine in the liver gives rise to N-methylated xanthines, methyluric acids, and acetylated uracil metabolites44. Using a targeted urinary metabolomics approach for coffee and caffeine-related compounds, we observed that the NCD group excreted lower levels of 1- and 3-methylxanthine, theobromine, and several methyluric acids compared with CD (Supplementary Fig. 15). Unsurprisingly, these metabolites are all associated with the metabolism of caffeine. Beyond caffeine-related metabolites, CDs also showed higher urinary levels of phenolic metabolites in comparison with NCD, including caffeoylquinic, cinnamic, phenylpropanoic, phenylacetic, and benzoic acids as well as hydroxybenzene derivatives. This is fully aligned with the high contribution of coffee (poly)phenols to the overall excretion of phenolic metabolites.
Following the period of caffeine abstinence, CDs urinary metabolite profile almost mirrored that of NCDs at the start of the study, including both alkaloids and phenolic metabolites. Exceptions were theobromine, 3-methyxanthine and 3,7-dimethyl uric acid, which were not significantly changed compared to pre-washout levels. Upon the reintroduction of either caffeinated coffee or decaffeinated coffee, most phenolic metabolites increased reaching concentrations in line with the values recorded for CDs. However, caffeine metabolites returned to CD values only upon caffeinated consumption but not for decaffeinated coffee, as expected.
In general, data showed expected trends for caffeine-derived and phenolic-derived compounds from coffee, although different metabolites were more associated with coffee reintroduction than others, with some slight differences between the two types of coffee. Of note, a large inter-individual variability was noted for phenolic metabolites, which was much lower for alkaloids. This further supports a contributory role of the individual gut microbiota in the metabolism of coffee (poly)phenols (Supplementary Fig. 15).
Coffee intake drives targeted shifts in gut microbial populations without changing overall diversity
Coffee consumption influenced gut microbiota composition, with significant strain specific differences observed between groups (Fig. 3). In the faeces of NCDs, the strains Cryptobacterium curtum, Eggerthella sp. CAG:209, Eggerthela sp. Cag 51_9, and Firmicutes CAG:94 were significantly decreased compared with CD at baseline while Velonella parvula, Velonella sp. ACP1 and Haemophilus parainfluenzae were increased in NCD participants. During the period without coffee consumption, CDs showed reduced levels of Cryptobacterium curtum over 3 different timepoints during this period. Upon coffee reintroduction, significant alterations were observed in the gut microbiota composition, independent of caffeine content. Upon reintroduction of caffeinated or decaffeinated coffee, we observed a temporal shift in the levels of all strains identified as significantly altered. It is only on day 21 of the reintroduction phase that there was a significant “bloom” of Velonella sp. ACP1 in participants receiving caffeinated and decaffeinated coffee. Notably, Velonella parvula was significantly increased in the faeces of decaffeinated coffee drinkers, while not significantly increased in CD (Fig. 3).
Heatmap of microbial relative abundance relative to baseline coffee drinkers (CD). Horizontal bars within the same cell represent values of individual participants. For the coffee drinker subset, which features repeated measurements, measurements from the same participant are aligned on the y-axis. Red colour indicates positive effect size while blue reflects negative effect size, with intensity of red or blue corresponding to increased or decreased effect sizes respectively and white reflecting zero effect. Panels containing a text box with numbers reflects those comparisons with a Cohen’s d effect size of > 0 5 reflecting medium sized effects. Comparisons are NCD v CD, washout v CD baseline and CAF or DECAF v CD baseline values. Column one = species, column two = NCD, column three = CD, column four washout period for CD and column five reintroduction of caffeinated or decaffeinated coffee. n = 31 at baseline for CD and NCD, n = 31 during washout period, n = 15 during reintroduction of decaffeinated coffee and n = 16 during reintroduction of caffeinated coffee.
Alpha-diversity was significantly different between NCD and CD but was not significantly different when the diversity of CD at baseline was compared with post-washout or post-intervention, suggesting that the influence of coffee or one of its constituent chemicals affects specific strains rather than overall diversity (Supplementary Data 16).
Caffeinated and decaffeinated coffee influence total and specific (poly)phenol consumption
We quantified daily (poly)phenol content using the food intake data from the 7-day food diaries provided by participants. Comparing CD with NCD, total (poly)phenols were higher in CD, largely reflecting higher levels of phenolic acids, particularly hydroxycinnamic acids (Supplementary Data 17a). Following a 2-week period of coffee abstinence, total (poly)phenols were decreased in CD, primarily due to a decrease in phenolic acids, especially hydroxycinnamic acids (Supplementary Data 17b). When we estimated (poly)phenol intake and compared values at the end of the period of coffee abstinence with the end of the intervention period where participants received caffeinated or decaffeinated coffee there was an increase in both groups in total (poly)phenols, phenolic acids and hydroxycinnamic acids, whereas other (poly)phenols increased only in the caffeinated coffee group (Supplementary Data S17,c).
Coffee-induced metabolic and microbial changes correlate with cognitive and behavioral outcomes
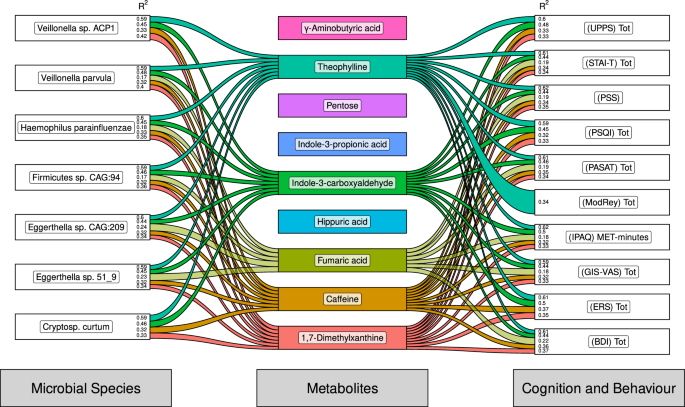
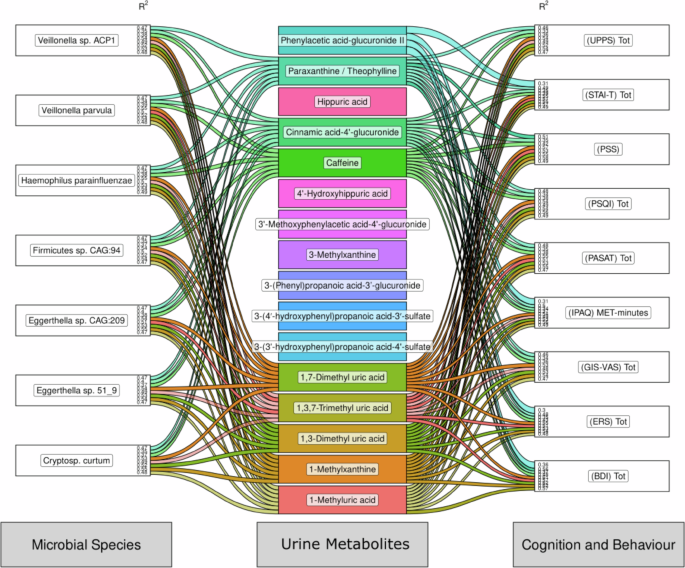
The gut microbiome is understood to influence host metabolism, as well as its own metabolic potential. Indeed, the microbiome and metabolites are capable of influencing cognition and behaviour. To investigate the interplay between the cognitive, metabolic, and microbial changes we have shown to be influenced by coffee consumption (and abstinence) we performed pairwise mixed model analysis between cognitive scores, microbial species abundance and faecal metabolite concentrations (Fig. 4). We see that both Velonella species are strongly associated with theophylline that in turn is strongly associated with several cognitive scores, including PSS, PSQI and ModRey, amongst others. Caffeine itself is associated with all species listed and is correlated with all cognitive and behavioral outputs. This data suggests that both metabolites and species are responsive to the presence of coffee and may influence anxiety.
Sankey diagram summarising pairwise statistical associations between microbial species (left), metabolites (centre) and cognition and behaviour (right) based on the integration of data from Fig. 1, 2 and 3. Each node represents a species, metabolite, or behavioural or cognitive output. Each line connecting a node represents a statistical association between that microbial species, metabolite, and cognitive output, as measured with a generalised linear mixed-effects model. The left (microbial species) and right (cognition and behaviour) nodes contain R2 values that represent the determination coefficient between that column and a centre node (metabolites). The thickness of the connecting line represents the marginal, not conditional, R2 value of that particular model, scaled by the total R2 of associations with that feature. Lines are opaque if R2 > 0.3 and translucent if 0.3 > R2 > 0.1. Only marginal associations with Benjamini-Hochberg adjusted p-values of < 0.001 are shown.
Phenolic acid metabolites in feces and urine reflect coffee-derived dietary signatures independent of caffeine
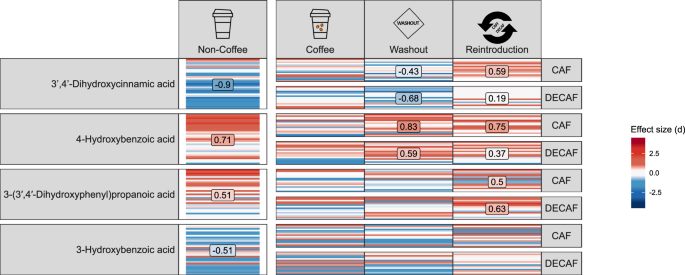
We quantified benzoic, phenylacetic, propanoic and cinnamic acid derivatives in faeces across the 3 distinct phases of the study. In NCDs, 3-hydroxybenzoic acid and 3ʹ,4ʹ-dihydroxycinnamic acid were lower compared to CDs, while 4-hydroxybenzoic acid and 3-(3ʹ,4ʹ-dihydroxyphenyl)propanoic acid were higher in NCDs (Fig. 5). Following the 2-week abstinence period, the level of 3ʹ,4ʹ-dihydroxycinnamic acid in CDs was significantly reduced, in line with the decrease of its coffee-derived precursors in the diet of participants, while 4-hydroxybenzoic acid was increased. Following the reintroduction of caffeinated or decaffeinated coffee there was no visible difference in metabolite concentration between caffeinated and decaffeinated coffee, with the levels of 3ʹ,4ʹ-dihydroxycinnamic acid, 4-hydroxybenzoic acid and 3-(3ʹ,4ʹ-dihydroxyphenyl)propanoic acid significantly increased compared with their baseline levels in all participants, regardless of caffeinated or decaffeinated coffee consumption.
Heatmap of targeted metabolomics relative to baseline coffee drinkers (CD). Horizontal bars within the same cell represent values of individual participants. For the coffee drinker subset, which features repeated measurements, measurements from the same participant are aligned on the y-axis. Red colour indicates positive effect size while blue reflects negative effect size, with intensity of red or blue corresponding to increased or decreased effect sizes respectively and white reflecting zero effect. Panels containing a text box with numbers reflects those comparisons with a Cohen’s d effect size of > 0. 5 reflecting medium sized effects. Comparisons are NCD v CD, washout v CD baseline and CAF or DECAF v CD baseline values. Column one = species, column two = NCD, column three = CD, column four washout period for CD and column five reintroduction of caffeinated or decaffeinated coffee. n = 31 at baseline for CD and NCD, n = 31 during washout period, n = 15 during reintroduction of decaffeinated coffee and n = 16 during reintroduction of caffeinated coffee.
Performing the same analysis on targeted (poly)phenol metabolites in the urine of participants we see further evidence of the collective influence of microbial species and members of the uric acid, xanthine, and glucuronide families of metabolites, that in turn are strongly associated with cognitive outputs (Fig. 6).
Sankey diagram summarising pairwise statistical associations between microbial species (left), metabolites (centre) and cognition and behaviour (right) based on the integration of data from Fig. 1, 2 and 3. Each node represents a species, metabolite, or behavioural or cognitive output. Each line connecting a node represents a statistical association between that microbial species, metabolite, and cognitive output, as measured with a generalised linear mixed-effects model. The left (microbial species) and right (cognition and behaviour) nodes contain R2 values that represent the determination coefficient between that column and a centre node (metabolites). The thickness of the connecting line represents the marginal, not conditional, R2 value of that particular model, scaled by the total R2 of associations with that feature. Lines are opaque if R2 > 0.3 and translucent if 0.3 > R2 > 0.1. Only marginal associations with Benjamini-Hochberg adjusted p-values of < 0.001 are shown.
Discussion
This study comprehensively investigated the effects of coffee consumption on cognition, mood, behavior, and gut microbiota in healthy adults, comparing moderate coffee drinkers to non-drinkers. It also explored changes during a 2-week coffee abstinence period and subsequent reintroduction with either caffeinated or decaffeinated coffee to distinguish caffeine and coffee effects. Utilizing shotgun metagenomics and targeted and untargeted metabolomics, this study furthers our understanding of how coffee influences the microbiota-gut-brain axis, including how it modulates stress, inflammation, cognition, and mood.
Comparing regular coffee drinkers with non-coffee drinkers revealed that coffee drinkers exhibited higher impulsivity and emotional reactivity. Attention and vigilance increased while impulsivity and emotional reactivity decreased upon withdrawal. Behaviourally, returning to caffeinated or decaffeinated coffee decreased perceived stress and self-reported symptoms of depression. Similarly, across both groups, impulsivity was also reduced following the reintroduction of caffeine or decaffeinated coffee. Interestingly, only reintroduction of caffeinated coffee resulted in a reduction in anxiety and psychological distress, confirming specific effects of caffeine on cognition and mood which has been reported by other groups45.
Further investigation into coffee’s impact on stress coping capacity included an examination of the stress hormone cortisol. Assessing the hypothalamic-pituitary-adrenal (HPA) axis function and cortisol production showed that the CAR and cortisol levels before, during, and after the SECPT were similar between coffee drinkers and non-coffee drinkers. This suggests that caffeine or coffee did not affect resilience to physical and social stress in either group. Other studies have suggested that coffee consumption activates cortisol production13,46, and it is worth noting that our study design did not specifically examine the acute effects of coffee on cortisol. It has also been shown that prolonged coffee consumption can diminish the cortisol response to stress, suggesting that habitual coffee drinkers may develop partial tolerance to stress13,15. Questionnaire results before and after the SECPT were consistent across both groups for anxiety, positive and negative affect, stress, and pain perception.
Given the observed effects of caffeine and coffee on emotional reactivity we sought to investigate whether immune function was involved in these changes. Coffee drinkers showed lower basal plasma levels of the acute-phase protein CRP and higher levels of the anti-inflammatory cytokine IL-10. Following exvivo stimulation of whole blood with LPS, coffee drinkers secreted significantly less IL-6 than non-coffee drinkers. These findings suggest that coffee or its components may have specific effects on immune function, potentially mediated by phenolic compounds with anti-inflammatory properties. Following the withdrawal of coffee, CRP levels increased alongside TNFα. There are conflicting findings on the impact of coffee on CRP levels; while some studies suggest an inverse association between black coffee consumption and high CRP levels, others indicate that coffee consumption is linked to lower CRP levels, suggesting potential anti-inflammatory properties47,48. Interestingly, IL-6 levels increased following LPS stimulation, providing consistent evidence of coffee’s influence on cytokine secretion. Further evidence of caffeine-specific effects was observed in cytokines, with IL-10 and IL-6 both reduced in the plasma of participants upon the reintroduction of caffeine but not those on decaffeinated coffee. In both caffeine and decaffeinated participants, LPS-induced IL-6 was halved on reintroduction of caffeine and decaffeinated coffee. Interestingly, increased dietary (poly)phenol intake has been shown to result in lower CRP levels49 suggesting a potential mechanism for the use of coffee in dietary-induced modification of gut-brain health.
We used untargeted metabolomic analysis of faecal material to explore the functional consequences of coffee on the host. Metabolomics revealed changes in coffee-associated compounds like caffeine and fumaric acid in coffee drinkers. Notably, neuroactive compounds such as GABA and IPA, were reduced in coffee drinkers. A recent study has shown that IPA plays a critical role in improving cognition in the elderly through dietary supplementation50. Further work should also explore the role of GABA and coffee, given both chemicals ability to influence anxiety51. ICA was also altered: ICA is a gut microbe derived tryptophan metabolite that has been suggested to influence gut barrier integrity and regulate gut homeostasis cooperatively via the cytokine IL-10 and the aryl hydrocarbon receptor (AHR)52. Targeted metabolomics of caffeine-related compounds revealed a clear link between coffee consumption and metabolic patterns, showing decreased levels of 3-hydroxybenzoic acid in the NCD group, while 4-hydroxybenzoic acid levels increased. Notably, abstinence from coffee and its reintroduction had a marked impact on these metabolite concentrations also.
Coffee drinkers exhibit distinct differences in their gut microbiome compared to non-coffee drinkers, with changes in α-diversity observed. These alterations suggest that coffee affects the gut microbiome independently of diet. Among coffee drinkers, specific species such as Cryptobacterium curtum, Eggertella sp, and Firmicutes bacterium are increased. Information on Cryptobacterium curtum and Eggertella CAG:209 are limited, with Cryptobacterium curtum associated predominately with oral health53, while the latter may be involved in bile acid synthesis54, aligning with coffee’s impact on acid secretion. Firmicutes CAG:94 has associations with positive emotions in females55. Although further research is needed to elucidate the specific functions of these strains, these findings suggest a positive influence of coffee on gut health. Coffee abstinence did not influence microbiome diversity with neither α nor β diversity were changed (Supplementary Data 16). At the species level, Eggerthella sp. CAG:209 and Firmicutes CAG:94 returned to levels similar to NCDs after abstinence, suggesting a direct influence by coffee or one of its components (Fig. 3.).
Examining the microbiome in those reintroducing coffee or decaffeinated coffee, neither α- or β-diversity is changed by coffee reintroduction at Day 2 of the intervention phase of the study. Interestingly, all seven of the strains different between NCD and CD were significantly affected by the reintroduction of either caffeinated or decaffeinated coffee suggesting that some of these strains may be sensitive to the presence of coffee or one of its constituent chemicals. Several of the differentially abundant species found in this study are normally resident in the oral cavity or part of the salivary or dental microbiome, further studies should dissect the potential of oral microbes to influence gut health given that the oral cavity is an endogenous reservoir for the gut microbiota56. The multiple bioactive features of coffee and caffeine should be examined further to determine the role they may play in coffee as a potential functional food or as a dietary supplement in disorders of the microbiome-gut-brain-axis.
Investigating the intricate interplay among coffee consumption, cognition, mood, immunity, and the gut microbiota in healthy adults illuminated multifaceted relationships influenced by caffeine. Integration of microbial, metabolic, and cognitive datasets provided a clear demonstration of how each integrates with the other. We see that of the nine metabolites changed, theophylline, ICA, fumaric acid, caffeine and 1,7-dimethylxanthine are highly connected to multiple microbial species and cognitive or behavioral outcomes while Firmicutes sp. CAG:94 and Eggerthella sp. 51_9 have significant connections with several metabolites indicating that these species have a strong influence on metabolic processes. Of particular interest we also see that GABA, pentose, IPA and hippuric acid are altered independent of microbial species or cognitive or behavioral output.
(Poly)phenols are widely consumed in the diet, with coffee being a major source of these compounds, which have been associated with beneficial cardiometabolic effects57,58. Leveraging sensitive, targeted metabolomics, we profiled the phenolic acid metabolism of our participants in both urine and faeces. Urinary data showed a clear profile between caffeine-derived metabolites, which declined during washout and were restored by caffeinated coffee, and (poly)phenol-derived metabolites, which were mostly elevated after both caffeinated and decaffeinated coffee. In contrast, fecal profiles reflected mainly (poly)phenol metabolism, with no distinction between the caffeinated and decaffeinated groups. The response to (poly)phenol consumption can be quite heterogeneous due to the presence or absence of specific genetic phenotypes of metabolic enzymes, gut microbiome composition, sex, age and current medications29,30,59. In fact, a high inter-individual variability was observed for the measured phenolics in urine, and it is increasingly acknowledged that the gut microbiome may influence the inter-individual variation in phenolic catabolism. Reviewing the (poly)phenol intake for all participants from detailed food diaries at distinct stages of the study, our data aligns with this hypothesis. We once again see the divergent influence that caffeinated coffee and decaffeinated coffee may have on (poly)phenol intake. These findings underscore the importance of considering both microbiome composition and beverage choice in future investigations of (poly)phenol metabolism and personalized nutrition strategies37,60.
It should also be noted that, overall, NCD participants appear to benefit from lower impulsivity and emotional reactivity, better cognitive stability, reduced inflammation risk, more stable blood pressure, distinct and potentially healthier gut microbiota and metabolite profiles and avoidance of caffeine withdrawal symptoms. These advantages may stem from the absence of a stimulant such as caffeine, allowing for the preservation of endogenous neurophysiological rhythms and homeostatic regulatory mechanisms, thereby promoting greater stability in cognitive, metabolic, and immunological function.
That said caffeinated coffee reduced anxiety, psychological distress, blood pressure, and improved attention and stress coping, while decaffeinated coffee enhanced sleep, physical activity, and memory. Both types lowered stress, depression, impulsivity, and inflammation, while also boosting mood and cognitive performance. Coffee influenced the gut microbiome, increased beneficial (poly)phenols and metabolites, and provided anti-inflammatory effects, suggesting that coffee, regardless of caffeine content, supports cognitive, psychological, immune, and metabolic health in distinct but complementary ways.
One limitation of our study is the absence of direct measurements of stool transit time which can be modulated by coffee. Given the invasive nature of such tests and their potential to alter the microbiome, we opted instead for surrogate markers—namely, the Bristol Stool Scale (BSS) and gastrointestinal visual analogue scale (GI-VAS)—which we believe are reasonable alternatives. However, we acknowledge that future studies investigating the impact of coffee on the gut microbiome should incorporate direct measures of stool transit61. We also recognize that baseline caffeine withdrawal in the NCD group may have influenced group differences. Nevertheless, the double-blinded crossover design helps mitigate this concern, and the level of caffeine omitted was likely below the threshold required to elicit withdrawal symptoms62. While GI-VAS has not been validated in healthy individuals, we consider its use appropriate within the context of our study. Additionally, we did not analyze differences by ethnicity due to the limited representation of ethnic groups in our sample (Table 1). Lastly, the study may not have been sufficiently powered to detect small to medium effect sizes for outcomes beyond our primary hypothesis—that coffee consumption influences the gut microbiome.
Through cutting-edge methodologies, this study unearthed distinctive effects of coffee on emotional reactivity, immune responses, and microbial composition, showcasing its potential as a modulator of the microbiota-gut-brain axis. Notably, periods of coffee abstinence highlighted enduring impacts on mood, immune markers, and microbial diversity, suggesting caffeine or other coffee constituents sustained influence. Moreover, the reintroduction of coffee, particularly caffeinated variants, revealed nuanced effects on stress perception, mood, and inflammatory markers, emphasizing caffeine-specific influences. We have demonstrated in granular detail the inter-individual metabolic profiles of phenolic compounds following coffee consumption and aligned this with the microbiome. We have sought to address causality between food or beverage consumption and the faecal microbiome, while also harnessing multi-omics integration. These findings not only illustrate the immediate microbial and neurotransmitter responses to caffeine but also emphasise the long-term implications for metabolite production within the microbiome. The study’s comprehensive insights pave the way for future investigations harnessing these interactions for potential health interventions and underscore the importance of understanding coffee’s multifaceted effects on human physiology.
Methods
Ethical approval
This protocol is a basic experimental study involving humans since it is a systematic study directed toward greater understanding on how coffee intake in healthy individuals influences the microbiome and behaviour with no intention to change the health status of the participants.
The study protocol was approved by the Clinical Research Ethic Committee of the Cork Teaching Hospitals (Study Identification Number APC115) and was registered in the Clinical Trials portal with the ClinicalTrials.gov ID NCT05927038, “Coffee Consumption, the Gut Microbiome, and the Microbiota-Gut-Brain Axis” and NCT05927103 “Differences Between Coffee and Non-coffee Drinkers in the Gut Microbiome and Microbiota-Gut-Brain Axis”. Informed consent was obtained from every participant enrolled in the study.
Study design
Sixty-two healthy adults between 30 and 50 years of age living in Ireland were recruited for the study between September 2021 and January 2023. Thirty-one participants were non-coffee drinkers (NCD, i.e., people that never consume coffee) and thirty-one participants were moderate coffee-drinkers (CD, i.e., people that usually consume between 3 to 5 cups of coffee per day). NCD and CD participants were compared cross-sectionally at baseline. CD participants only proceeded further with the study and were asked to abstain from any kind of coffee for 2 weeks. After the coffee washout period, CDs were block randomized (block of five, stratified by gender) into either caffeinated or decaffeinated coffee consumption group using a double-blinded, parallel design by the first author. Figure S17 details information relative to participants enrolment and allocation.
Study visits and coffee intervention
A scheme of the experimental design is displayed in Fig. 1A. During the screening visit at University College Cork the eligibility of the participants was verified. Demographics, medical and family history information were collected. MINI-international Neuropsychiatric Interview (M.I.N.I, version 7.0.2), Childhood Trauma Questionnaire (CTQ) and Rome IV Diagnostic Questionnaire for Functional Gastrointestinal Disorders in Adults (ROME-IV) were administered to assess the overall mental and gastrointestinal health of the participants63,64. Habitual caffeine consumption was assessed by a 7-day caffeine consumption diary and a brief measure of verbal IQ was completed using National Adult Reading Test (NART)65 (Supplementary Data 1). Both NCD and CD participants were asked to abstain from any other kind of caffeinated drink and dark chocolate during the week before the baseline visit, except their habitual coffee for CDs only.
During the baseline visit, the participants underwent a brief physical examination, and their samples were collected. Subsequently, the participants completed self-reported mood and behaviour questionnaires (see below), cognitive tasks and the Socially Evaluated Cold Pressor Test (SECPT). At the end of the baseline visit, NCDs participant concluded the study and received thirty euros compensation for any travel expenses incurred in taking part in the study. CDs participants were instructed to refrain from any caffeinated drink, any kind of coffee or dark chocolate for 2 weeks. After coffee/caffeine washout, CDs attended the pre-intervention visit in which they underwent a physical examination, samples collection and completed self-reported questionnaires as well as Paced Auditory Serial Additional Test (PASAT) cognitive test. During this visit, CDs were instructed to drink four sachets per day of the provided coffee (Nescafé Classic caffeinated or decaffeinated, 1.8 g instant coffee per sachet) for 3 weeks, beginning on that day.
No other coffee or caffeinated drinks were allowed during the intervention. The coffee provided was consumed with a quantity of hot water, milk, sugar chosen by the participant. CDs completed the study with the post-intervention visit (Visit 4), in which they completed the same measures as for V2 and received sixty euros compensation. Extra stool samples from CDs were collected 2 (day 2) and 4 (day 4) days after the start of coffee withdrawal and 2 (day 16), 4 (day 18) and 14 (day 28) days after the start of coffee intervention. Together with the samples, CDs completed mood, fatigue, and cravings questionnaires.
Exclusion criteria
The participants were instructed not to change dietary and food supplements habits. Participants were excluded from the study in the following situations: if affected by significant acute or chronic coexisting illness, if under any medication (except contraceptive pills and hormonal supplements) and antibiotics/probiotics/prebiotics (minimum 4 weeks of washout to participate), if vegan, if habitual consumer of high quantity of fermented foods (5-6 servings per day), if hypertensive, if pregnant or planning a pregnancy or lactating, if not fluent in English, if dyslexic/dyscalculic, if a current smoker, if involved in other experimental drugs or food trials with other companies or laboratory and if already participated as a volunteer in the Microbiota-Gut-Brain Axis lab (APC Microbiome Ireland) in the past 4 years.
Dietary intake quantification
Dietary intake was monitored and quantified by using a 7-day food diary66. The food diary was completed three times, before the baseline visit, before the pre-intervention visits and before the post-intervention visit. Each day, every participant had to note anything they eat or drink throughout the day, specifying the amount/portion size, the method of preparation/brand and comments. The Nutritics tool (https://www.nutritics.com/en/) was used to enter the food diary data and to quantify the macronutrients consumed. The 7-day food diary was a paper-based document that was completed at the timepoints mentioned. It included instructions how to estimate portion size, how to describe the method of preparation and how the food was consumed. The information was then manually entered into NutriticsTM. Food intake was weighted.
Dietary intake of (poly)phenols
A total of 1424 food items were retrieved from the food diaries. Foods not containing (poly)phenols (n = 391) were excluded from this analysis. All (poly)phenol-containing raw foods were univocally matched with items collected in an in-house database based on Phenol-Explorer 3.667 (www.phenol-explorer.eu). Raw food items that could not be associated to any item in Phenol-Explorer were associated with the most similar food or sourced through multiple literature searches and added to the in-house database. Recipes or complex foods were analysed by considering the individual ingredients and their relative quantity in the food. Specifically, recipes from the food diaries were used and, when not available, an online search was conducted. Changes in weight during cooking or processing were adjusted using yield factors from Bognar’s tables68, Phenol explorer69, or CREA70 (e.g., cooked food, fruit juice, and espresso) wherever relevant.
Microsoft Access was used to match the food composition table with the one related to food intakes, and to calculate (poly)phenol intake by multiplying the amount of food consumed by the (poly)phenol content expressed in mg/100 g. All available compounds belonging to flavonoid, phenolic acid, lignan and other (poly)phenol classes of interest were considered, including total (poly)phenol content assessed through the Folin assay method. Total (poly)phenol content was also calculated as the sum of individual phenolics determined by chromatography without hydrolysis, or by chromatography after hydrolysis if non-hydrolyzed chromatographic values were unavailable, across all considered compound classes except for a few (poly)phenols where the data after hydrolysis is more reliable (as for lignans). Additionally, normal-phase high-performance liquid chromatography (HPLC) data were used for proanthocyanidins, while individual data obtained by reverse-phase HPLC were used when the previous one was not available.
Daily average intakes were analyzed both as total classes (flavonoids, phenolic acids, lignans, and others) and subclasses (anthocyanins, flavan-3-ols, flavanones, flavones, flavonols, isoflavones, hydroxybenzoic acids, hydroxycinnamic acids, hydroxyphenylacetic acids, hydroxyphenylpropanoic acids, chalcones, ellagitannins, prenylflavonoids, avenanthramides, stilbenes and several miscellaneous minor (poly)phenols across study periods.
Lastly, the main dietary sources of (poly)phenolic compounds were also calculated for total (poly)phenol intake and for each phenolic class. Dietary sources were analyzed as food groups as well as subgroups or specific food items (Supplementary Data S,17-S,29).
Caffeine consumption quantification
During the screening visit, both NCD and CD participants completed a 7-day caffeine consumption diary. In the diary, each participant reported all the caffeinated beverages consumed in the previous 7 days. For each day, they reported the kind of caffeinated drinks consumed (e.g., instant coffee, filtered coffee, cappuccino, flat white, black tea, energy drink), the amount consumed (in cups, or cans or bottles in ml and how many e.g., 3 240 ml mugs) and the method of preparation, the brand, or any other known useful information about a specific beverage. Based on the specific caffeinated beverages consumed by the participants, a table with caffeine content of the drinks was created (Supplementary Data 1) and was used to calculate the average of daily caffeine intake, daily caffeine intake from coffee only and daily caffeine amount given up for the study, all expressed in mg of caffeine/day.
Self-reported questionnaires
During visits 2, 3 and 4
Every participant completed the following questionnaires: Cohen’s Perceived Stress Scale (PSS)71, Emotional and Reactivity Scale (ERS)72, Urgency Premeditation Perseverance Sensation Seeking Positive Urgency Impulsive Behaviour Scale (UPPS-P)73, Hopkins Symptom Checklist 90 (SCL 90-R)74, Beck Depression Inventory-II (BDI)75, State Trait Anxiety Inventory (STAI, during SECPT, State was used, and during baseline, post-withdrawal and post—intervention Trait was used)76, Pittsburgh Sleep Quality Index (PSQI)77, International Physical Activity Questionnaire (IPAQ)78, Gastrointestinal Symptoms Visual Analogue Scales (GI-VAS). These questionnaires aimed to assess the level of stress, emotional reactivity, impulsivity, depression, sleep quality, physical activity gastrointestinal symptoms. Stool types were reported using the Bristol stool chart (BSC)61.
During washout and intervention
At the timepoints (T2W, T4W, T2I, T14I) during coffee withdrawal and intervention periods as well as during visit 3 and 4, CD participants were asked to complete the following questionnaires to monitor caffeine cravings and fatigue: Caffeine Withdrawal Symptom Questionnaire (CWSQ)79, Questionnaire of Caffeine Cravings (QCC)80 and Visual Analogue Scale to evaluate Fatigue severity (VAS-F)81.
Neuropsychological assessment
Verbal list learning and episodic memory
Modified Rey Auditory Verbal Learning Test (ModRey). The ModRey was administered to estimate the participant’s episodic memory. Participants learned two lists of words (A and B) each consisting of twenty words. Participants had to recall the words at three moments: immediately, following a short delay (5 min) and after a long delay (30 min), followed by a recognition task.
Word list A was played aloud via a standardised audio recording, after which the participant recalled as many words as possible (immediate recall). This process was completed two more times. After which, a new list of words (list B) was introduced, and the participant was again asked to immediately recall this word list. 5 min later (short delay), participants were asked to recall words from List A. After 30 min (long delay), participants again recalled words from both List A and B.
A recognition phase was then performed in which participants had to indicate whether each presented word came from list A, B, or neither of those lists82. To prevent learning effects, separate word lists were used for baseline and endpoint visits.
Visuospatial memory and new learning
Paired Associates Learning (PAL) from Cambridge Neuropsychological Test Automated Battery (CANTAB) included eight levels with increasing difficulty. White boxes were displayed on the screen and revealed in sequence. Some boxes would be empty, and others would reveal a pattern which the participants had to memorize. Once the content of every box was revealed, all the previously shown patterns would appear in the centre of the screen, one by one. For each pattern appearing, the participants had to touch the screen to select the box in which the pattern had originally appeared. In the case of an error, the sequence was shown again. The main outcome measure was the total number of errors summed across the entire task. This test does not include different versions of forms; thus, it cannot be repeated too frequently to avoid learning bias. Due to this limitation, this test was administered during visit 2 and visit 4 and not during visit 3.
Emotional processing
Emotion Recognition Task (ERT) from CANTAB has been used to assess emotional processing, which is often associated with depression and affective disorders. ERT measures the ability to identify six basic emotions in facial expressions along a continuum of expression magnitude (Happiness, Anger, Surprise, Sadness, Fear, Disgust). Consecutively, one of the six emotions computer-morphed images were displayed for 200 milliseconds on the centre of the screen. The subject had to then select amongst the six options which emotion had been shown. This test lasted between 6 to 10 min. Outcome measures include numbers of correct or incorrect answers and overall response latencies in individual emotions and across all emotions. This test does not include different versions of forms; thus, it cannot be repeated too frequently to avoid learning bias. Due to this limitation, this test was administered during visit 2 and visit 4 and not during visit 3.
Attention and vigilance
Paced Auditory Serial Additional Test (PASAT) included an audio recording where single digit numbers played. Participants were asked to sum the two most recent digits spoken on the tape. PASAT is composed by two trials83. The single digit numbers (60 numbers each trial) were spoken at a rate one every 3 s (3’) and 2 s (2’) respectively for each trial. The PASAT outputs included the percentages of correct answers for each trial, 3’and 2’, as well as the total percentage of correct answers.
PASAT was performed during visit 2, 3 and 4 since it has limited learning bias, and two different forms are available. Form A was administered during visits 2 and 4, and form B was administered during visit 3. Form A and Form B differ in the numbers used on each 3’ and 2’ trial.
Socially evaluated cold pressor test
The Socially Evaluated Cold Pressor Test (SECPT) is a psychological and physical stressor that was used to induce an acute stress response by the activation of the hypothalamus-pituitary-adrenal axis (HPA axis)84. The participants were led into a room different from the one used in study visit one, in which a researcher was present, a video camera and a bucket with water containing ice (0 °C). Each participant had to sit facing the camera and the researcher and was told that their facial expression would be recorded for later analysis and that the researcher was recording the non-verbal behaviour. Then, they were asked to submerge their hand, including the wrist, in the ice-cold water for three minutes unless the participant indicated they could no longer continue. During those three minutes, each participant was instructed to stare at the camera. At the end of the procedure, the participant was given a paper towel to dry their hand and allowed to leave the room.
Before and after the SECPT, eight saliva samples were collected as detailed in the methods section and the participants had to complete different stress reported questionnaires as an indication of anxiety, stress, mood, and pain related to the stress procedure only.
Prior to the SECPT, each participant completed the following questionnaires: STAI-State (Ferreira & Murray 1983), Primary Appraisal Secondary Appraisal (PASA)85, Positive Negative Affect Schedule PANAS86, Bond-Lader Visual Analogue Scales (BL-VAS)87 and level of stress felt before the procedure (0% to 100%, VAS stress). After SECPT, each participant completed the following questionnaires: STAI-State, VAS stress after the procedure, VAS stress during the procedure, difficulty to keep the hand in the water (0% to 100%) and how unpleasant was the SECPT (0% to 100%), PANAS, BL-VAS and level of pain felt during the SECPT (1 to 10, VAS pain).
Biological samples
Biological samples were collected on the day of Visit 2 (V2), Visit 3 (V3) and Visit 4 (V4) and analysed as follows.
Saliva
The morning of the visit, participants were asked to collect four saliva samples. The first sample was collected within 3 minutes after awakening, the second 30 min later, and the third and fourth 45 min and 60 min respectively after wakening. These were used to measure the concentration of the cortisol awakening response (CAR) which gives an indication of HPA axis function46,88. In addition, participants were asked to collect eight saliva samples during the SECPT in the order indicated in Fig. S15.These samples were used to assess the short-term release of cortisol before, during and after the stress procedure.
Saliva samples were collected using Salivette Cortisol tubes (Sarstedt) and kept refrigerated until delivery to the laboratory. The saliva tubes were centrifuged at g for 5 min at 4 °C. The obtained saliva was stored at −80 °C until analysis.
Blood
Blood samples were collected from the antecubital vein during each visit. Whole blood was collected into 4 ml lithium-heparin-containing tubes (Vacuette, Greiner Bio-One, 454029). Blood for plasma samples was collected into 4 ml K2E K2EDTA tubes (Vacuette, Greiner Bio-One, 454023). Blood for serum samples was collected into 3.5-ml CAT SERUM Sep Clot Activator tubes (Vacuette, Greiner Bio-One, 454071). Blood for serum samples was incubated for 20 min at room temperature and blood for plasma samples was stored at 4 °C. Afterwards, blood samples were centrifuged at 1500 × g for 10 min at 4 °C to obtain serum and plasma. Both were aliquoted and stored at −80 °C.
An aliquot of 1 mL of whole blood was diluted 1:10 in medium (DMEM, 10% Foetal Calf Serum (FCS) and 5% Pen strep). A 500 µL aliquot of diluted blood was stimulated with 5 µL of LPS-EK -TLR4 agonist, lipopolysaccharide from E. coli K12 (Human TLR1-9 Agonist Kit, InvivoGen) and incubated for 24 h at 37 °C. Stimulated and unstimulated whole blood was aliquoted and stored at −80 °C.
Urine
The first urine of the day was collected and kept refrigerated in a sterile collection tube by each participant, and 4 ml of urine were aliquoted into 1 ml of 0.25% sodium azide and stored at −80 °C.
Stool
Participants were asked to collect the first freshly voided stool samples of the day in plastic containers containing an AnaeroGen sachet (Oxoid AGS AnaeroGen Compact, Fischer Scientific, Dublin). Stool samples were refrigerated using ice packs until delivery to the laboratory, where they were aliquoted and stored at −80 °C. Stool samples were collected the day of the visits as well as during the extra timepoints during the coffee washout and coffee intervention.
Biological samples analysis
Cortisol analysis
Cortisol concentration within the saliva samples was measured using Cortisol enzyme-linked immunosorbent assay (ELISA) kit (Enzo Life Sciences, #ADI-901-071, Exeter, UK) according to manufacturer’s instructions. Saliva samples were diluted 1:3 in the provided assay buffer (200 μL of Assay Buffer + 100 μL of saliva sample). The absorbance was measured with Synergy (BioTek) plate reader and Gen5 software, with a lower detection limit of 57 pg/mL. Each saliva sample was tested in duplicate, and the average of each concentration was used to determine the final concentration of cortisol. The area under the curve was calculated88.
Inflammatory markers analysis
Inflammatory markers were analysed both in whole blood (LPS-stimulated and unstimulated) and in plasma. Concentration of tumour necrosis factor alpha (TNFα), interleukin-1 beta (IL-1β), interleukin-6 (IL-6), interleukin-8 (IL-8), interleukin-10 (IL-10), and interferon gamma (IFNγ) was determined by using V-PLEX Proinflammatory Panel 1 (human) Kit MSD®MULTI-SPOT assay system (Meso Scale Discovery, USA) in either plasma or whole blood or both, according to the manufacturer’s protocol. No samples dilution was required. The concentration of C-reactive protein was determined by using V-PLEX Vascular Injury Panel 2 (human) Kit MSD®MULTI-SPOT assay system (Meso Scale Discovery, USA) in plasma samples diluted 1:1000 with diluent 100 and 101, following the manufacturer’s instructions. Cytokine concentrations were calculated using a 4-parameter logistical curve within the MSD DISCOVERY Methodological Mind Software. Results for each cytokine were expressed in pg/mL. C-reactive protein (CRP) was converted to mg/L. The results are shown as normalised concentration of cytokines in the whole blood. The normalized concentration was calculated by subtracting the unstimulated whole blood concentration from the LPS-stimulated whole blood concentration. This calculation was performed to better account for inter-individual differences.
Faecal gut microbiota analysis
DNA was extracted from 250–500 mg of sample using the QIAamp Power Faecal Pro Kit from Qiagen with homogenisation on a Tissuelyser (Qiagen) at 30 Hz for 10 min. DNA was quantified using the Qubit Broad Range Assay (Invitrogen) and 200–300 ng were used as input for shotgun library preparation. Samples were prepared using Illumina DNA prep (Illumina). Following library prep, samples were pooled at equal concentrations. The average size of the DNA fragments was determined using the Agilent High Sensitivity kit (Agilent) and concentration was determined using the Qubit High Sensitivity Assay. The final pool was then sequenced on a NovaSeq 6000 S2 flow cell according to the manufacturer’s instructions.
Microbiome: taxonomic and functional analysis
We performed quality checks on raw sequences from all faecal samples using the FastQC program. Shotgun metagenomic sequencing data were then cleaned, and host genome sequences were filtered using Bowtie289 via the Kneaddata wrapper program with following parameters: ILLUMINACLIP:/NexteraPE-PE.fa:2:30:10, SLIDINGWINDOW:5:25, MINLEN:60, LEADING:3, TRAILING:3. Reads were aligned to the Web of Life database using Bowtie2 and taxonomic and functional profiling of the microbial community was performed using woltka90. Next, the uniref90-based gene abundance matrix was further collapsed by KEGG Orthology (KO) term mapping via the “woltka tools collapse” function provided within woltka. Woltka SOP is available online (https://github.com/qiyunzhu/woltka/blob/master/doc/wolsop.sh). Gut-Brain Modules (GBMs) and Gut-Metabolic Modules (GMMs) were calculated using the R version of the Gomixer tool91.
Metabolomics
Semi-polar metabolite analysis was carried out by MS-Omics as follows. The analysis was carried out using a Thermo Scientific Vanquish LC coupled to a Orbitrap Exploris 240 MS, Thermo Fisher Scientific. An electrospray ionization interface was used as ionization source. Analysis was performed in positive and negative ionization mode under polarity switching. The UPLC was performed using a slightly modified version of the protocol described by Doneanu et al. (UPLC/MS Monitoring of Water-Soluble Vitamin Bs in Cell Culture Media in Minutes, Water Application note 2011, 720004042en). Peak areas were extracted using Compound Discoverer 3.3 (Thermo Scientific). Identification of compounds were performed at four levels; Level 1: identification by retention times (compared against in-house authentic standards), accurate mass (with an accepted deviation of 3ppm), and MS/MS spectra, Level 2a: identification by retention times (compared against in-house authentic standards), accurate mass (with an accepted deviation of 3ppm). Level 2b: identification by accurate mass (with an accepted deviation of 3ppm), and MS/MS spectra, Level 3: identification by accurate mass alone (with an accepted deviation of 3ppm).
SCFA analysis was carried out by MS-Omics as follows. Samples were acidified using hydrochloride acid, and deuterium labelled internal standards where added. All samples were analyzed in a randomized order. Analysis was performed using a high polarity column (Zebron™ ZB-FFAP, GC Cap. Column 30 m x 0.25 mm × 0.25 μm) installed in a GC (7890B, Agilent) coupled with a quadropole detector (5977B, Agilent). The system was controlled by ChemStation (Agilent). Raw data was converted to netCDF format using Chemstation (Agilent), before the data was imported and processed in Matlab R2021b (Mathworks, Inc.) using the PARADISe software described by Johnsen et. al (DOI: 10.1016/j.chroma.2017.04.052).
Urine sample preparation for targeted metabolomics
For phenolic analysis, urine samples were defrosted, vortexed, diluted in 0.1% formic acid in water (1:5 v/v), centrifuged at 17,968 × g for 5 min, and filtered (0.22 μm nylon filter)26. For caffeine and caffeine metabolites, a subsequent dilution in acidified water was performed (1:50 v/v). For pyridine analysis, 10 µL of urine mixed with pure acetonitrile (1:100 v/v), vortexed for 1 min, and centrifuged at 13 765 × g for 10 min at room27. For creatinine, urine samples were centrifuged at 10,980 g for 5 min and subsequently diluted with water 0.1% formic acid (1:1000 v/v), vortexed, and transferred into vials for analysis28.
UPLC-ESI-QqQ-MS/MS analysis for phenolics, caffeine, and caffeine metabolites:
An Ultra-high performance liquid chromatography (UHPLC) Acquity UHPLC I-Class Plus system (Waters Corporation, Milford, MA, USA) equipped with a binary pump, autosampler, and oven with an Acquity Premier HSS T3 column (1.8 µm, 2.1 × 100 mm i.d., Waters) installed with an Acquity VanGuard Premier HSS T3 1.8 um (2.1 × 5 mm) precolumn were used. The injection volume was 5 µL, the samples were maintained at 10 °C, and the column at 40 °C. The mobile phase consisted of water with 0.01% formic acid (A) and acetonitrile (ACN) with 0.01% formic acid (B). A gradient elution was applied as follows: 0 min, 99% (A); 0.5 min, 99% (A); 3.0 min, 85% (A); 6.0 min, 50% (A), 9.0 min, 5% (A), 10.0 min, 5% (A), 11.0 min, 99% (A); 14.0 min, 99% (A). A flow rate of 400 µL/min was applied. A Xevo XS triple-quadrupole mass spectrometer with an electrospray ionization source (UHPLC-ESI-QqQ-MS/MS, Waters) operating in negative and positive modes was used. The capillary voltage was set at 2.3 kV. The source temperature was set to 150 °C, and the desolvation gas to 600 °C. The gas flows were set to cone gas 150 L/h, desolvation gas 800 L/h and nebulizer at 7.0 L/h. The mass spectrometer operated in multiple reaction monitoring (MRM) mode and cone, and collision voltage were optimized for each compound by infusion experiments with standards. When not available, the energies were optimized in chromatography or used from molecules with similar structures. Equipment control and data acquisition were performed using MassLynx software version 4.2 and data processing by TargetLynx Software (Waters). A total of 125 urine (n = 1) samples were analyzed,
UHPLC-ESI-QqQ-MS/MS analysis for pyridines (trigonelline derivatives):
Samples were analyzed using a UHPLC DIONEX Ultimate 3000 fitted with a TSQ Vantage triple quadrupole mass spectrometer equipped with a heated-ESI source (Thermo Fisher Scientific Inc., San Jose, CA, USA)25,26. Separations were performed using XBridge® BEH HILIC column (2.1 × 100 mm; 2.5 µm particle size; Waters) installed with a precolumn cartridge (Waters). Volume injection was 5 µL and column oven was set to 35 °C. Flow rate was set at 0.5 mL/min, mobile phase A was water containing 0.1% formic acid, mobile phase B was acetonitrile containing 0.1% formic acid, and mobile phase C was ammonium formate 20 mM containing 0.1% formic acid. For mobile phase C, the proportion of 10% was kept throughout the run. Following 0.5 min of 1% A in 89% of B, the proportion of B was decreased to 80% for a period of 3 min. Solvent B was decreased again to 47% for 1.5 min and increased back to 89% from 6.5 min to the end of the run (total run: 13 min). The HESI-II source interface was set to a capillary temperature of 270 °C and the source heater temperature was 200 °C. The sheath gas pressure was set at 40 (arbitrary units), the auxiliary gas pressure at 5 (a.u.), and the source voltage was 3.5 kV. A total of 3 compounds were monitored through selective reaction monitoring (SRM) mode. Quantification was performed with calibration curves of authentic standards. Data processing was performed using Xcalibur software version 2.1 (Thermo Scientific Inc., Waltham, MA, USA). A total of 125 urine samples (n = 1) were analysed (Supplementary Data 22).
UHPLC-ESI--MS/MS analysis for creatinine:
Analysis was performed with an UHPLC Accela 1250 equipped with a linear ion trap-mass spectrometer (LTQ XL) fitted with a heated-ESI probe (Thermo Fisher Scientific) (Tosi et al., 2023). Separations were performed using XSelect HSS T3 column (50 × 2.1 mm; 2.5 µm particle size; Waters) installed with a precolumn cartridge (Phenomenex, CA, USA). Volume injection was 5 µL and column oven was set to 40 °C. Flow rate was set at 0.2 mL/min, mobile phase A was water containing 0.1% formic acid and mobile phase B was acetonitrile containing 0.1% formic acid. Chromatographic separation was performed in an isocratic mode keeping A and B at 50% for 2 min. The HESI-II interface was set to a capillary temperature of 275 °C and the source heater temperature was 300 °C. The sheath gas (N2) flow rate was set at 40 (a.u.) and the auxiliary gas (N2) flow rate at 5 a.u. The source voltage was 4.5 kV, and the capillary and tube lens voltage were +20 and +95 V, respectively. Ultra-high purity helium gas and a CID equal to 55 were used to obtain MS2 fragmentation. Creatinine was analyzed with a full-scan MS2 analysis in a positive ionization mode, monitoring all the ions with a m/z value between 35 and 200 Da. Chromatograms and spectral data were acquired using XCalibur software 2.1 (Thermo Fisher Scientific). Identification and quantification were performed with calibration curves built with the available standard. A total of 125 urine samples (n = 1) were analysed.
Targeted metabolomics of faecal samples
Faecal samples were thawed, weighed, proportionally diluted in ethyl acetate acidified (1200 µL for every 300 mg of sample) with formic acid (0.1%), vortexed, and sonicated for 10 min at room temperature29,30. Vortexing and sonication (5 min) were repeated once before centrifuging (14000 rpm, 10 min, 4 °C). The supernatant was recovered, and the pellet was re-extracted following the same procedure. The resulting supernatants were pooled, vortexed, dried in a vacuum concentrator, resuspended in 200 μL of MeOH: H2O (50:50, v/v acidified with formic acid (0.1%)), sonicated, centrifuged, and filtered (0.22 μm nylon) into vials for UHPLC-ESI-QqQ-MS/MS analysis.
Faecal extracts were analyzed using the same instrument described for pyridine analysis. Separation was carried out by means of a Kinetex EVO C18 column (100 × 2.1 mm; 2.6 µm particle size; Phenomenex), installed with a precolumn cartridge (Phenomenex) (Di Pede et al., 2022). Mobile phase, pumped at a flow rate of 0.4 mL/min, consisted of a mixture of acidified water (0.01% v/v formic acid) (solvent A) and acidified acetonitrile (0.01% v/v formic acid) (solvent B). Following 0.5 min of 5% solvent B in A, the proportion of B was increased linearly to 40% over a period of 7 min. Solvent B was increased again to 80% in 1 min, maintained for 2 min and then the start conditions were re-established in 0.5 min and maintained for 3 min to re-equilibrate the column (total run: 14 min). The HESI-II source interface was set to a capillary temperature of 275 °C and the source heater temperature was 250 °C. The sheath gas (N2) flow rate was set at 40 a.u., the auxiliary gas (N2) flow rate at 5 a.u. and the sweep gas flow was set at 15 a.u. The source voltage was 3 kV; the capillary voltage was −9 V and tube lens voltage was −53 V. A total of 52 compounds were monitored through SRM mode. Quantification was performed with calibration curves of standards, when available, or using the most structurally similar compound. Data processing was performed using Xcalibur software version 2.1. A total of 123 faecal samples (n = 1) were analysed once. Quality controls were performed using one point of the calibration curve during the batch for every 30 samples (Supplementary Data 23).
Target metabolomics quality assessment
For all targeted metabolomics analyses, metabolites were identified by matching retention times and fragmentation patterns with authentic standards. When standards were not available, characteristic fragments of the corresponding phenolic class were monitored and cross-checked with both the in-house library and literature references. Peak quality was evaluated based on peak shape, and peaks deviating by more than 0.05 minutes from the reference were excluded. Quality control was ensured by injecting a calibration-curve point every 30 samples to monitor potential instrumental drift and sensitivity loss. Calibration curves included at least seven points, with linearity values above r = 0.98. Accuracy for each point ranged between 80% and 120%. The limit of quantification was defined as the first calibration point with a signal-to-noise ratio above 10 and meeting the linearity criteria. Final metabolite concentrations were expressed relative to creatinine for urine samples and per unit weight (mg/g) for faecal samples. The validated dataset was then used for all subsequent analyses. The targeted metabolomics data have been deposited to MetaboLights repository with the study identifier MTBLS1349492 (Supplementary Data 22 and 23)
Adenosine A2A receptor (ADORA2A) genotyping
DNA was extracted from 100 µl of whole blood using the DNeasy Blood&Tissue kit (Qiagen). Two Single Nucleotide Polymorphisms (SNPs) related to caffeine sensitivity were selected using the data base https://www.ensembl.org/index.html and previous scientific evidence40,93,94,95,96. The SNPs selected are rs5751876 (Frequency MAF: 0.44. synonymous variant) and rs2298383 (Frequency MAF: 0.40, non-coding transcript exon variant). DNA was diluted to obtain a concentration of 4 ng/µl in 50 µl of elution buffer.
SNPs genotyping analysis of rs2298383 and rs5751876 was performed at Translational Analysis in Molecular Medicine (TAMM) at Karolinska University Hospital (Huddinge, Sweden) using iPLEX® Gold chemistry and the MassARRAY® mass spectrometry system97 (Agena Bioscience, San Diego, CA, U.S.A.). A two-plexed assay was designed using MassARRAY® Assay Design Suite v3.0 software (Agena Bioscience), genotyping the two SNPs in one reaction per sample. The protocol for allele-specific base extension was performed according to Agena Bioscience’s recommendation. Analytes were spotted onto a 384-element SpectroCHIP II array (Agena Bioscience) using Nanodispenser RS1000 (Agena Bioscience) and subsequently analysed by MALDI-TOF on a MassARRAY® Analyzer 4 mass spectrometer (Agena Bioscience). Genotype calls were manually checked by two persons individually using MassARRAY® TYPER v5.0 Software (Agena Bioscience).
The genotyping assay was validated using a set of fourteen trio families, in total forty-two individuals, with genotype data available for rs2298383 through the HapMap consortium for39 individuals, and for rs5751876 through the 1000 Genomes Project Phase 3 for 21 individuals, respectively. Concordance analysis with the available data showed 100% concordant genotypes for both SNPs. Parent-offspring-compatibility analysis was performed, and no Mendel error was observed in the genotyped trio families for any of the SNPs. Three individual control samples were genotyped in parallel with both the validation sample run and the run of the study samples. All control samples showed 100% concordant genotypes for both SNPs. Additionally, genotyping the study samples in duplicate resulted in 100% concordant genotypes for both SNPs.
Bioinformatics
Further data-handling was undertaken in R (version 4.2.0) using the Rstudio GUI (version 2022.2.2.485). In all microbiome analysis except for alpha diversity, taxa with a prevalence of <60% of samples at the species level were excluded from analysis as ratios are invariant to subsetting and this study employs compositional data analysis techniques. Principal component analysis was performed on centred log-ratio transformed (clr) values. Zeroes were replaced using the ‘const’ approach described by Lubbe and colleagues. Beta diversity was computed in terms of Aitchison distance, or Euclidean distance between clr-transformed data, and assessed by PERMANOVA using the vegan package. Alpha diversity was computed using the iNEXT library. Differential abundance of microbial features and metabolites was assessed using linear models, or linear mixed effect models in the case of longitudinal (repeated measures) data. Variables were selected based on the Benjamini-Hochberg procedure with a q-value of 0.2 as a threshold. This procedure controls the False Discovery Rate (FDR) using sequential modified Bonferroni correction for multiple testing. Figures were generated in R, using ggplot2 and its extensions patchwork and ggforce.
Statistical analysis
Our primary outcome measure was microbiota composition and function (see NCT05927103) while secondary outcome measures included gut microbial metabolites (including SCFAs) and Coffee-related metabolites. Other secondary outcomes included cognitive performance, response to an acute stressor, peripheral blood inflammatory profile and the cortisol awakening response. g*Power was used for power calculations considering an α-error probability (false positive) of 0.05 and a power of 80%. This calculation indicated that the minimum required samples size per group was 18, thus, 36 participants were required for analysis. Effect sizes of 0.25 will be able to be differentiated using n = 18 per group (ANOVA repeated measures) for microbiota composition and function. Allowing for attrition, we estimated the requirement to recruit a minimum of 60 volunteers were recruited. Final recruitment numbers were 62, following screening of 118 potential participants and enrolment in the study of 92. Statistical analyses were performed using SPSS version 28 (IBM, Armonk, NY, USA) and n R (version 4.2.0) using the Rstudio GUI (version 2022.2.2.485). Outliers have been identified using the Grubb’s test (α = 0.05) and were removed before analysis. Every set of data except demographic data and (poly)phenols intake from food diaries were tested for outliers. Only statistical outliers were removed due to modest sample size used in the study. General Linear Model Univariate was used to perform statistical analysis of all the data of NCD versus CD in visit 2 (baseline), except CAR and SECPT timepoints as well as immediate versus delayed recall of List A in the ModRey. Age, gender, BMI, and amount of caffeine given up were used as covariates. All the other statistical analyses were performed using Mixed Models followed by Bonferroni’s post hoc pairwise comparisons at each timepoint. Statistical analysis for categorical data, such as mode of delivery and genotyping data, were performed using Pearson’s chi-squared test. p < 0.05 was deemed significant in all analysis. Data are expressed as mean + SEM unless otherwise stated.
Reporting summary
Further information on research design is available in the Nature Portfolio Reporting Summary linked to this article.
Data availability
The data that supports the findings of this study are deposited in the repositories listed below. The untargeted metabolomics data generated in this study has been deposited in the MetaboLights database under accession code MTBLS1340192. The targeted metabolomics data is available at MetaboLights under accession code MTBLS13494, [https://www.ebi.ac.uk/metabolights/MTBLS13494]. Counts for microbiome (10.5281/zenodo.18661295) are on Zenodo as is participant metadata, it has been uploaded to 10.5281/zenodo.18348935. The raw microbiome data generated in this study are provided in the ENA database under accession code PRJEB108545.
Code availability
All original code has been deposited at GitHub (https://github.com/thomazbastiaanssen/coffee/) and is publicly available.
References
de Melo Pereira G. V., et al. Chapter Three - Chemical composition and health properties of coffee and coffee by-products. In: Toldrá F, ed. Advances in Food and Nutrition Research. Vol 91. Academic Press; 2020:65-96.
Saud, S. & Salamatullah, A. M. Relationship between the Chemical Composition and the Biological Functions of Coffee. Mol. Basel Switz. 26, 7634 (2021).
Grosso, G., Godos, J., Galvano, F. & Giovannucci, E. L. Coffee, Caffeine, and Health Outcomes: An Umbrella Review. Annu Rev. Nutr. 37, 131–156 (2017).
Simon, J. et al. Light to moderate coffee consumption is associated with lower risk of death: a UK Biobank study. Eur. J. Prev. Cardiol. 29, 982–991 (2022).
Noyce, A. J. et al. Meta-analysis of early nonmotor features and risk factors for Parkinson disease. Ann. Neurol. 72, 893–901 (2012).
Godos, J. et al. Dietary (Poly)phenols and Cognitive Decline: A Systematic Review and Meta-Analysis of Observational Studies. Mol. Nutr. Food Res. 68, 2300472 (2024).
Qi, H. & Li, S. Dose-response meta-analysis on coffee, tea and caffeine consumption with risk of Parkinson’s disease. Geriatr. Gerontol. Int 14, 430–439 (2014).
Wang, L., Shen, X., Wu, Y. & Zhang, D. Coffee and caffeine consumption and depression: A meta-analysis of observational studies. Aust. N. Z. J. Psychiatry 50, 228–242 (2016).
Poole, R. et al. Coffee consumption and health: umbrella review of meta-analyses of multiple health outcomes. BMJ 359, j5024 (2017).
Liu, Q. P. et al. Habitual coffee consumption and risk of cognitive decline/dementia: A systematic review and meta-analysis of prospective cohort studies. Nutr. Burbank Los Angel Cty Calif. 32, 628–636 (2016).
Magalhães, R. et al. Habitual coffee drinkers display a distinct pattern of brain functional connectivity. Mol. Psychiatry 26, 6589–6598 (2021).
Li J., Yu K., Bu F., Li P., Hao L. Exploring the impact of coffee consumption and caffeine intake on cognitive performance in older adults: a comprehensive analysis using NHANES data and gene correlation analysis. Nutr. J. 2025;24. https://doi.org/10.1186/s12937-025-01173-x
Lovallo, W. R. et al. Caffeine stimulation of cortisol secretion across the waking hours in relation to caffeine intake levels. Psychosom. Med. 67, 734–739 (2005).
Fiani, B. et al. The Neurophysiology of Caffeine as a Central Nervous System Stimulant and the Resultant Effects on Cognitive Function. Cureus 13, e15032 (2021).
MacKenzie, T. et al. Metabolic and hormonal effects of caffeine: randomized, double-blind, placebo-controlled crossover trial. Metabolism 56, 1694–1698 (2007).
Papakonstantinou, E. et al. Acute effects of coffee consumption on self-reported gastrointestinal symptoms, blood pressure and stress indices in healthy individuals. Nutr. J. 15, 26 (2016).
Iriondo-DeHond, A., Uranga, J. A., Del Castillo, M. D. & Abalo, R. Effects of Coffee and Its Components on the Gastrointestinal Tract and the Brain-Gut Axis. Nutrients 13, 88 (2020).
Nehlig, A. Effects of Coffee on the Gastro-Intestinal Tract: A Narrative Review and Literature Update. Nutrients 14, 399 (2022).
Prebiotic potential of coffee and coffee by-products. In: Coffee in Health and Disease Prevention. Elsevier; 2025:817-827.
Cowan, T. E. et al. Chronic coffee consumption in the diet-induced obese rat: impact on gut microbiota and serum metabolomics. J. Nutr. Biochem. 25, 489–495 (2014).
Pérez-Burillo, S., Rajakaruna, S., Pastoriza, S., Paliy, O. & Ángel Rufián-Henares, J. Bioactivity of food melanoidins is mediated by gut microbiota. Food Chem. 316, 126309 (2020).
Dalile, B., Van Oudenhove, L., Vervliet, B. & Verbeke, K. The role of short-chain fatty acids in microbiota–gut–brain communication. Nat. Rev. Gastroenterol. Hepatol. 16, 461–478 (2019).
González, S. et al. Long-Term Coffee Consumption is Associated with Fecal Microbial Composition in Humans. Nutrients 12, 1287 (2020).
Harakeh, S. et al. Impact of smoking cessation, coffee and bread consumption on the intestinal microbial composition among Saudis: A cross-sectional study. PloS One 15, e0230895 (2020).
Jaquet, M., Rochat, I., Moulin, J., Cavin, C. & Bibiloni, R. Impact of coffee consumption on the gut microbiota: a human volunteer study. Int J. Food Microbiol. 130, 117–121 (2009).
Mills, C. E. et al. In vitro colonic metabolism of coffee and chlorogenic acid results in selective changes in human faecal microbiota growth. Br. J. Nutr. 113, 1220–1227 (2015).
Nakayama, T. & Oishi, K. Influence of coffee (Coffea arabica) and galacto-oligosaccharide consumption on intestinal microbiota and the host responses. FEMS Microbiol Lett. 343, 161–168 (2013).
Sales, A. L. et al. Effects of regular and decaffeinated roasted coffee (Coffea arabica and Coffea canephora) extracts and bioactive compounds on in vitro probiotic bacterial growth. Food Funct. 11, 1410–1424 (2020).
Favari, C. et al. Factors driving the inter-individual variability in the metabolism and bioavailability of (poly)phenolic metabolites: A systematic review of human studies. Redox Biol. 71, 103095 (2024).
Di Pede, G. et al. A Systematic Review and Comprehensive Evaluation of Human Intervention Studies to Unravel the Bioavailability of Hydroxycinnamic Acids. Antioxid. Redox Signal 40, 510–541 (2024).
Madeira, M. H., Boia, R., Ambrósio, A. F. & Santiago, A. R. Having a Coffee Break: The Impact of Caffeine Consumption on Microglia-Mediated Inflammation in Neurodegenerative Diseases. Mediators Inflamm. 2017, 4761081 (2017).
Muqaku, B. et al. Coffee consumption modulates inflammatory processes in an individual fashion. Mol. Nutr. Food Res 60, 2529–2541 (2016).
Zampelas, A., Panagiotakos, D. B., Pitsavos, C., Chrysohoou, C. & Stefanadis, C. Associations between coffee consumption and inflammatory markers in healthy persons: the ATTICA study. Am. J. Clin. Nutr. 80, 862–867 (2004).
Chatterjee, A. et al. Dietary polyphenols represent a phytotherapeutic alternative for gut dysbiosis associated neurodegeneration: A systematic review. J. Nutr. Biochem 129, 109622 (2024).
Asnicar, F. et al. Microbiome connections with host metabolism and habitual diet from 1,098 deeply phenotyped individuals. Nat. Med. 27, 321–332 (2021).
Manghi, P. et al. Coffee consumption is associated with intestinal Lawsonibacter asaccharolyticus abundance and prevalence across multiple cohorts. Nat. Microbiol. 9, 3120–3134 (2024).
Schneider, E., O’Riordan, K. J., Clarke, G. & Cryan, J. F. Feeding gut microbes to nourish the brain: unravelling the diet–microbiota–gut–brain axis. Nat. Metab. 6, 1454–1478 (2024).
Cryan, J. F. et al. The Microbiota-Gut-Brain Axis. Physiol. Rev. 99, 1877–2013 (2019).
Zeng, L., Xiang, R., Fu, C., Qu, Z. & Liu, C. The Regulatory effect of chlorogenic acid on gut-brain function and its mechanism: A systematic review. Biomed. Pharmacother. 149, 112831 (2022).
Rétey, J. V. et al. A Genetic Variation in the Adenosine A2A Receptor Gene (ADORA2A) Contributes to Individual Sensitivity to Caffeine Effects on Sleep. Clin. Pharm. Ther. 81, 692–698 (2007).
Berres, S. & Erdfelder, E. The sleep benefit in episodic memory: An integrative review and a meta-analysis. Psychol. Bull. 147, 1309–1353 (2021).
Moore, D. & Loprinzi, P. D. Exercise influences episodic memory via changes in hippocampal neurocircuitry and long-term potentiation. Eur. J. Neurosci. 54, 6960–6971 (2021).
Clifford, M. N. et al. Exploring and disentangling the production of potentially bioactive phenolic catabolites from dietary (poly)phenols, phenylalanine, tyrosine and catecholamines. Redox Biol. 71, 103068 (2024).
Martínez-López, S. et al. Theobromine, caffeine, and theophylline metabolites in human plasma and urine after consumption of soluble cocoa products with different methylxanthine contents. Food Res Int 63, 446–455 (2014).
Kim, H. et al. Drinking coffee enhances neurocognitive function by reorganizing brain functional connectivity. Sci. Rep. 11, 14381 (2021).
Harris, A., Ursin, H., Murison, R. & Eriksen, H. R. Coffee, stress and cortisol in nursing staff. Psychoneuroendocrinology 32, 322–330 (2007).
Choi, S. & Je, Y. Association between coffee consumption and high C-reactive protein levels in Korean adults. Br. J. Nutr. 130, 2146–2154 (2023).
Zhang, Y. & Zhang, D. Z. Is coffee consumption associated with a lower level of serum C-reactive protein? A meta-analysis of observational studies. Int J. Food Sci. Nutr. 69, 985–994 (2018).
Dryer-Beers E. R., Griffin J., Matthews P. M., Frost G. S. Higher Dietary Polyphenol Intake Is Associated With Lower Blood Inflammatory Markers. J Nutr. Published online May 11, 2024:S0022-316600282-00287. https://doi.org/10.1016/j.tjnut.2024.05.005
Kim, C. S., Jung, S., Hwang, G. S. & Shin, D. M. Gut microbiota indole-3-propionic acid mediates neuroprotective effect of probiotic consumption in healthy elderly: A randomized, double-blind, placebo-controlled, multicenter trial and in vitro study. Clin. Nutr. 42, 1025–1033 (2023).
Nehlig, A., Daval, J. L. & Debry, G. Caffeine and the central nervous system: mechanisms of action, biochemical, metabolic and psychostimulant effects. Brain Res Rev. 17, 139–170 (1992).
Powell, D. N. et al. Indoles from the commensal microbiota act via the AHR and IL-10 to tune the cellular composition of the colonic epithelium during aging. Proc. Natl. Acad. Sci. USA 117, 21519–21526 (2020).
Lopez-Oliva, I. et al. Dysbiotic Subgingival Microbial Communities in Periodontally Healthy Patients With Rheumatoid Arthritis. Arthritis Rheumatol. Hoboken NJ 70, 1008–1013 (2018).
Doden, H. L. et al. Completion of the gut microbial epi-bile acid pathway. Gut Microbes 13, 1907271 (2021).
Ke, S. et al. Gut feelings: associations of emotions and emotion regulation with the gut microbiome in women. Psychol. Med. 53, 7151–7160 (2023).
Schmidt, T. S. et al. Extensive transmission of microbes along the gastrointestinal tract. eLife 8, e42693 (2019).
Ziauddeen, N. et al. Dietary intake of (poly)phenols in children and adults: cross-sectional analysis of UK National Diet and Nutrition Survey Rolling Programme (2008–2014). Eur. J. Nutr. 58, 3183–3198 (2019).
Tresserra-Rimbau, A. et al. Inverse association between habitual polyphenol intake and incidence of cardiovascular events in the PREDIMED study. Nutr. Metab. Cardiovasc Dis. 24, 639–647 (2014).
Morand, C. et al. Why interindividual variation in response to consumption of plant food bioactives matters for future personalised nutrition. Proc. Nutr. Soc. 79, 225–235 (2020).
Sanz, Y. et al. The gut microbiome connects nutrition and human health. Nat. Rev. Gastroenterol. Hepatol. 22, 534–555 (2025).
Lewis, S. J. & Heaton, K. W. Stool Form Scale as a Useful Guide to Intestinal Transit Time. Scand. J. Gastroenterol. 32, 920–924 (1997).
Juliano, L. M. & Griffiths, R. R. A critical review of caffeine withdrawal: empirical validation of symptoms and signs, incidence, severity, and associated features. Psychopharmacol. (Berl.) 176, 1–29 (2004).
Bernstein, D. P. et al. Initial reliability and validity of a new retrospective measure of child abuse and neglect. Am. J. Psychiatry 151, 1132–1136 (1994).
Schmulson, M. J. & Drossman, D. A. What Is New in Rome IV. J. Neurogastroenterol. Motil. 23, 151–163 (2017).
Nelson H., Willison J. %J. W. ON, Canada. National Adult Reading Test (NART) Nfer-Nelson. Published online 1991.
Harrington, J. et al. Sociodemographic, health and lifestyle predictors of poor diets. Public Health Nutr. 14, 2166–2175 (2011).
Neveu, V. et al. Phenol-Explorer: an online comprehensive database on polyphenol contents in foods. Database 2010, bap024–bap024 (2010).
Bognár A. Tables on Weight Yield of Food and Retention Factors of Food Constituents for the Calculation of Nutrient Composition of Cooked Foods (Dishes). BFE; 2002. https://books.google.ie/books?id=KvYOSwAACAAJ
Rothwell, J. A. et al. Phenol-Explorer 3.0: a major update of the Phenol-Explorer database to incorporate data on the effects of food processing on polyphenol content. Database 2013, bat070–bat070 (2013).
AlimentiNUTrizione - Presentazione Dati. Accessed December 2, 2024. https://www.alimentinutrizione.it/presentazione-dati
Cohen, S., Kamarck, T. & Mermelstein, R. A global measure of perceived stress. J. Health Soc. Behav. 24, 385–396 (1983).
Nock, M. K., Wedig, M. M., Holmberg, E. B. & Hooley, J. M. The emotion reactivity scale: development, evaluation, and relation to self-injurious thoughts and behaviors. Behav. Ther. 39, 107–116 (2008).
Cyders, M. A., Littlefield, A. K., Coffey, S. & Karyadi, K. A. Examination of a short English version of the UPPS-P Impulsive Behavior Scale. Addict. Behav. 39, 1372–1376 (2014).
Lipman, R. S., Covi, L. & Shapiro, A. K. The Hopkins Symptom Checklist (HSCL)-factors derived from the HSCL-90. J. Affect Disord. 1, 9–24 (1979).
Arnarson, T. O., Olason, D. T. & Smári, J. Sigurethsson JF. The Beck Depression Inventory Second Edition (BDI-II): psychometric properties in Icelandic student and patient populations. Nord J. Psychiatry 62, 360–365 (2008).
Ferreira, R. & Murray, J. Spielberger’s State-Trait Anxiety Inventory: Measuring anxiety with and without an audience during performance on a stabilometer. Percept. Mot. Skills 57, 15–18 (1983).
Buysse, D. J., Reynolds, C. F., Monk, T. H., Berman, S. R. & Kupfer, D. J. The Pittsburgh Sleep Quality Index: a new instrument for psychiatric practice and research. Psychiatry Res 28, 193–213 (1989).
Booth, M. Assessment of physical activity: an international perspective. Res. Q Exerc. Sport. 71, 114–120 (2000).
Juliano, L. M., Huntley, E. D., Harrell, P. T. & Westerman, A. T. Development of the caffeine withdrawal symptom questionnaire: caffeine withdrawal symptoms cluster into 7 factors. Drug Alcohol Depend. 124, 229–234 (2012).
West, O. & Roderique-Davies, G. Development and initial validation of a caffeine craving questionnaire. J. Psychopharmacol. Oxf. Engl. 22, 80–91 (2008).
Shahid A., Wilkinson K., Marcu S., Shapiro C. M. Visual Analogue Scale to Evaluate Fatigue Severity (VAS-F). In: Shahid A., Wilkinson K., Marcu S., Shapiro C. M., eds. STOP, THAT and One Hundred Other Sleep Scales. Springer; 2012:399-402.
Hale, C. et al. The ModRey: An Episodic Memory Test for Nonclinical and Preclinical Populations. Assessment 26, 1154–1161 (2019).
Correia S. Paced Auditory Serial Attention Test. In: Kreutzer J. S., DeLuca J., Caplan B., eds. Encyclopedia of Clinical Neuropsychology. Springer; 2011:1840-1845.
Schwabe, L., Haddad, L. & Schachinger, H. HPA axis activation by a socially evaluated cold-pressor test. Psychoneuroendocrinology 33, 890–895 (2008).
Gaab, J., Rohleder, N., Nater, U. M. & Ehlert, U. Psychological determinants of the cortisol stress response: The role of anticipatory cognitive appraisal. Psychoneuroendocrinology 30, 599–610 (2005).
Watson, D., Clark, L. A. & Tellegen, A. Development and validation of brief measures of positive and negative affect: the PANAS scales. J. Pers. Soc. Psychol. 54, 1063–1070 (1988).
Bond, A. & Lader, M. The use of analogue scales in rating subjective feelings. Br. J. Med. Psychol. 47, 211–218 (1974).
Pruessner, J. C. et al. Free cortisol levels after awakening: a reliable biological marker for the assessment of adrenocortical activity. Life Sci. 61, 2539–2549 (1997).
Langmead, B. & Salzberg, S. L. Fast gapped-read alignment with Bowtie 2. Nat. Methods 9, 357–359 (2012).
Zhu, Q. et al. Phylogeny-Aware Analysis of Metagenome Community Ecology Based on Matched Reference Genomes while Bypassing Taxonomy. Sharpton T. J., ed. mSystems 7, e00167–22 (2022).
Darzi, Y., Falony, G., Vieira-Silva, S. & Raes, J. Towards biome-specific analysis of meta-omics data. ISME J. 10, 1025–1028 (2016).
Yurekten, O. et al. MetaboLights: open data repository for metabolomics. Nucleic Acids Res. 52, D640–D646 (2024).
Alsene, K., Deckert, J., Sand, P. & de Wit, H. Association between A2a receptor gene polymorphisms and caffeine-induced anxiety. Neuropsychopharmacol. Publ. Am. Coll. Neuropsychopharmacol. 28, 1694–1702 (2003).
Banks, N. F. et al. Genetic Polymorphisms in ADORA2A and CYP1A2 Influence Caffeine’s Effect on Postprandial Glycaemia. Sci. Rep. 9, 10532 (2019).
Erblang, M. et al. The Impact of Genetic Variations in ADORA2A in the Association between Caffeine Consumption and Sleep. Genes 10, 1021 (2019).
Landolt, H. P. No Thanks, Coffee Keeps Me Awake”: Individual Caffeine Sensitivity Depends on ADORA2A Genotype. Sleep 35, 899–900 (2012).
Oeth P., et al. iPLEXTM Assay: Increased Plexing Efficiency and Flexibility for MassARRAY System Through Single Base Primer Extension with Mass-Modified Terminators. Published online January 1, 2005.
Acknowledgements
We are grateful to the Institute for Scientific Information on Coffee (ISIC) for funding this research. We would like to thank Aonghus Lavelle, David A. Lachmansingh, Gabriela Ribeiro, Jason Martin, David Kaulmann, Thieza Melo, Antonio Girella, and Eric Kelleher for their support. We also wish to thank all the study participants for their involvement in our study. We acknowledge the use of the APC Microbiome Ireland Flow Cytometry Platform. APC Microbiome Ireland is a research centre funded by Science Foundation Ireland (SFI), through the Irish Governments’ national development plan (grant no. 12/RC/2273_P2). This work was also partially supported by the European Research Council (ERC) under the European Union’s Horizon 2020 research and innovation programme (PREDICT-CARE project, grant agreement No 950050) and the National Recovery and Resilience Plan (NRRP), Mission 4 Component 2 Investment 1.3 - Call for tender No. 341 of March 15, 2022 of Italian Ministry of University and Research funded by the European Union – NextGenerationEU; Award Number: Project code PE00000003, Concession Decree No. 1550 of October 11, 2022 adopted by the Italian Ministry of University and Research, CUP D93C22000890001, Project title“ON Foods - Research and innovation network on food and nutrition Sustainability, Safety and Security – Working ON Foods”. Icons were used in the creation of some figures sourced from www.flaticon.com (Icon creator Freepik).
Ethics declarations
Competing interests
J.F.C. has been an invited speaker at conferences organised by Bromotech, Yakult and Nestle and has received research funding from Nutricia, DuPont/IFF, and Nestle. G.C. has received honoraria from Janssen, Probi, Apsen, and Ingelheim Boehringer as an invited speaker; is in receipt of research funding from Pharmavite, Fonterra, Reckitt, Nestle and Tate and Lyle; and has been paid for consultancy work by Yakult, Zentiva, Bayer Healthcare and Heel Pharmaceuticals. ES has received honorarium from Janssen Sciences Ireland UC. ES also received honorarium from MyNutriWeb for an event that was sponsored by Yakult. This support neither influenced nor constrained the contents of this manuscript. The remaining authors declare no competing interests.
Peer review
Peer review information
Nature Communications thanks Paul J Arciero, and the other, anonymous, reviewer for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Boscaini, S., Bastiaanssen, T.F.S., Moloney, G.M. et al. Habitual coffee intake shapes the gut microbiome and modifies host physiology and cognition. Nat Commun 17, 3439 (2026). https://doi.org/10.1038/s41467-026-71264-8
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41467-026-71264-8