Introduction
Hair loss, or alopecia, represents a widespread disease with significant psychological and social implications, affecting millions of people worldwide. Moreover, people suffered from hair loss is expected to rise due to aging and lifestyle-induced stress1. Conventional hair loss treatments primarily include pharmaceutical interventions, such as minoxidil and finasteride, and procedural approaches like hair transplants2,3. While these treatments have demonstrated adequate efficacy in hair growth, they are often accompanied by many drawbacks. Specifically, pharmaceutical treatments require prolonged use of medications and can cause side effects like irritation and hormonal imbalances, while hair transplants are invasive and costly4,5,6. In light of these potential risks, photobiomodulation (PBM) has emerged as a promising non-pharmacological and non-invasive alternative for hair regrowth, by stimulating cellular activity without experiencing side effects associated with pharmaceutical treatments7,8.
PBM offers considerable therapeutic potential across a range of biomedical applications, including hair-loss treatment. However, conventional PBM devices primarily utilize LED or laser-based platforms, each of which presents critical limitations for wearable use. LED-based systems often exhibit non-uniform light dispersion, leading to inconsistent dosimetry across the target area. Laser-based systems, while offering improved directionality, are typically rigid and bulky, which significantly restricts mobility and user comfort.
A notable class of commercial PBM systems for hair-loss treatment adopts helmet- or hairband-type configurations incorporating arrays of LEDs and lasers9,10,11. This device is characterized by a mechanically robust helmet-type design that can be securely fixed to the head. However, the platforms are restricted to use in indoor home environments rather than during daily outdoor activities, and the reliance on numerous point‑like LED and laser emitters, each illuminating only a small area, necessitates large emitter counts to cover the scalp, which can increase the overall device weight.
To address these limitations, wearable PBM platforms are required that deliver spatially uniform light stimulation without compromising user mobility. Recent studies have explored alternative light‑source–based wearables, including micro‑LED patches and OLED‑based cage devices. Micro‑LED patches offer a more flexible form factor than rigid helmet or home‑use phototherapy systems and can improve conformity to skin. However, limitations arise from the patch properties and the micro‑scale emitting area of individual sources. Because attachment typically relies on skin adhesives, patches can inadvertently induce hair traction during application and removal, which is a non‑trivial concern for patients with weakened hair follicle anchorage12. In addition, the pixelated point‑source nature of micro‑LEDs complicates scaling to large treatment areas and tends to produce non‑uniform irradiance with local hot spots, thereby limiting dose homogeneity across the scalp. Motivated by these constraints, OLED‑based phototherapy device has been suggested. Unlike conventional light sources for phototherapy device, such as LEDs and lasers, OLEDs offer a combination of ultra-thin, flexible form factors and uniform light emission. An OLED-based platform provides large, homogeneous emission and have been reported to induce PBM relevant to hair‑loss treatment13. However, barriers to day‑to‑day wearability persist. The platform has been designed for helmet-type configurations, and as a result, core OLED attributes such as thin profiles and mechanical flexibility remain underutilized.
Furthermore, the light irradiation should be capable of stimulating cellular activation. The PBM effect depends on the target cell type and wavelength band. Recent studies indicate that human dermal papilla cells (hDPCs), which play a crucial role in hair regeneration14, exhibit enhanced proliferation after exposure to near-infrared (NIR) irradiation compared with red light at the same dose, providing the importance of selecting an optimal wavelength for effective treatment15,16.
NIR light (700–900 nm) exhibits superior tissue penetration because strong absorption by melanin and water is avoided and scattering is reduced, enabling a range of biomedical applications7,17,18. These optical characteristics make NIR light particularly suitable for non-invasive phototherapy, allowing efficient delivery of therapeutic light to subdermal target19.
An NIR OLED, in which the PBM advantages of NIR light are coupled with the flexibility of OLEDs, is a promising light source for wearable phototherapy systems. Despite these promising characteristics, the practical implementation of wearable NIR OLEDs remains relatively unexplored. While some studies have demonstrated the fabrication of NIR OLEDs on flexible plastic substrates for wrist-worn sensor applications, the broader utilization of this technology beyond sensing remains uncommon20,21. One of the major limitations is the mechanical fragility of traditional NIR OLEDs, which have generally based on indium tin oxide (ITO) electrodes. Due to the brittle nature of ITO, these devices lack the mechanical resilience required for repeated bending and deformation, which is essential for true wearable applications.
Most NIR OLEDs reported to date adopt a bottom‑emitting structure, in which light exits through the substrate. An optical characteristic of the NIR OLEDs are highly dependent on substrate transmittance, which inherently restricts the range of applicable substrate materials15,22. These factors have hindered the adoption of NIR OLEDs in wearable biomedical applications, emphasizing the need for alternative device structures to enhance versatility in such applications.
In this study, we report a wearable textile-based NIR OLEDs that employ a microcavity structure to optimize light characteristics for enhanced human dermal papilla cell (hDPCs) activation for hair-loss treatment. The device consisted of around 240µm-thick polyester-based textile platform and a top-emitting NIR OLED combined with a flexible encapsulation layer to protect the OLED from oxygen and water, with a total thickness of less than 1 µm. The textile substrate provides a user-friendly, flexible platform suitable for integration into wearable items such as hats or clothing23,24,25.
To achieve wavelength-optimized operation of NIR OLEDs intended for hDPC-targeted phototherapy, a microcavity structure was implemented. This architecture enables spectral tuning without changing the emitter material, because PBM responses are dependent on wavelength. In this configuration, the conventional ITO electrode was replaced with a thin silver layer, forming a top-emitting microcavity device with improved optical characteristics and mechanical flexibility, which in turn enables controlled spectral tuning and use as a wearable device. By adjusting the organic thickness of the OLED, the emission peak can be precisely tuned in 10 nm increments within the NIR spectral range (700–800 nm) with sufficient light intensity for phototherapy, enabling the NIR OLED characteristics to be tailored specifically to match the action spectrum peak for optimal activation of hDPCs7.
The NIR OLED was integrated into a textile platform and demonstrated its feasibility as a skin-contact wearable device. The flexibility of the textile-based top-emitting OLEDs were validated through repeated bending tests, confirming stable radiance output and mechanical robustness without delamination even after 10,000 bending cycles at a bending radius of 2 mm, corresponding to 2.1% tensile strain. The device exhibited water resistance, retaining over 90% of its initial performance after 50 h of water immersion. Additionally, continuous operation of the devices for 20 min resulted in a surface temperature below 30 °C on the emitting side (the skin-contact area), demonstrating the device’s thermal safety against skin burns.
To validate the biological responses of the NIR OLED system, in vitro studies were conducted using hDPCs, focusing on cellular migration and senescence. As a result, improved migration activity was observed in hDPCs exposed to NIR OLED irradiation compared with non-irradiated controls. Furthermore, greater suppression of senescence-associated β-galactosidase activity and senescence markers was observed under irradiation with the NIR device than under red OLED and conventional NIR OLED conditions with a full-width at half-maximum (FWHM) greater than 100 nm. A 91.6% reduction in senescence-associated markers was demonstrated following exposure to the microcavity NIR OLED relative to the non-irradiated control group.
Results
Wearable NIR OLEDs for phototherapy
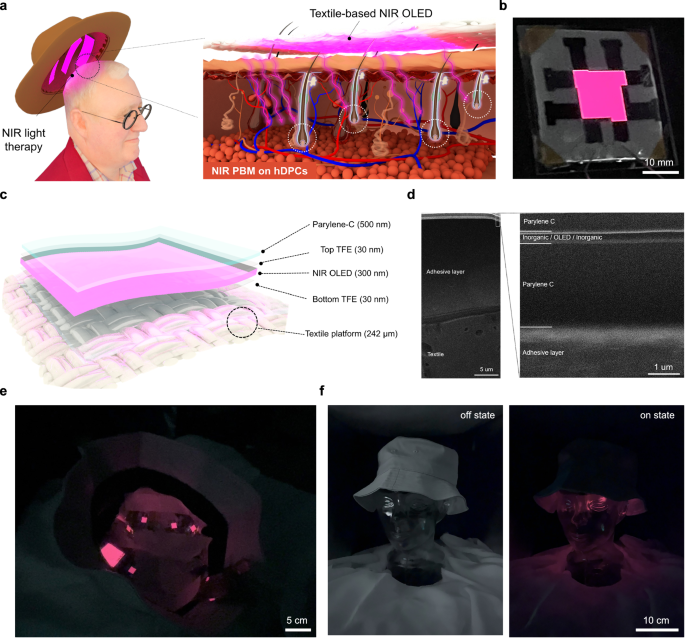
A device is designed for targeted photic activation of hDPCs using NIR illumination (Fig. 1a). The wearable textile-based NIR OLED is used as a non-invasive photobiomodulation (PBM) device for hair-loss treatment. Near-infrared emission from the textile device is transmitted through the skin to the dermal papilla, where human dermal papilla cells are activated.
a Schematic of the study concept in which near‑infrared OLED emission delivers photic stimulation to human dermal papilla cells. b Photograph of an operating textile‑based NIR OLED showing a spatially uniform, planar emitting surface. c Device architecture illustrating sequential formation of thin‑film encapsulation and NIR OLED stacks on a textile platform. The outermost parylene‑C functions as a skin‑contact barrier and as an optical capping layer. d Cryo‑FIB‑SEM cross‑section of the NIR OLED. The textile stack comprises a polyester substrate, an adhesive interlayer, and parylene‑C beneath the thin‑film encapsulation, OLED layers, and the outermost parylene‑C. e Textile‑based NIR OLED integrated into a commercial bucket hat. f Mannequin head fitted with the textile‑based NIR OLED in the off (left) and on (right) states.
The fabrication process of textile-based device entails the sequential deposition of functional layers onto an uncoated, pristine polyester textile substrate. Textile consists of bundles of fibrous filaments, ensuring flexibility and mechanical strength (Supplementary Fig. 1). In addition, the polyester-based textile, commonly used in clothing, offers excellent flexibility, making it highly suitable as a wearable platform (Supplementary Fig. 2a).
However, the regular textile weave introduces substantial surface roughness, which can compromise the performance of OLEDs fabricated by thermal deposition. Given the intrinsic roughness associated with a ~ 170 µm weave, planarization was applied to enable NIR OLED fabrication and to ensure stable driving. To achieve a uniform surface, a 3 µm parylene-C layer was deposited via chemical vapor deposition (CVD) and mechanically bonded to the textile substrate using a 19 µm adhesive layer26. This process significantly reduced surface roughness, ensuring uniform deposition of materials in giving stable NIR OLED operation (Fig. 1b).
To shield the OLED devices from moisture and oxygen, a bottom thin-film encapsulation (TFE) layer composed of Al2O3/TiO2 nanolaminates (30 nm) was deposited via atomic layer deposition (ALD) at 40 °C. This method preserved the mechanical integrity of the polyester textile27. Subsequently, both OLED electrodes and organic layers were fabricated using physical vapor deposition (PVD) to establish a top-emitting structure, where light can be propagated in the direction opposite to the textile-based platform.
The polyester exhibited a poor transmittance of only 21% in the visible-to-NIR range (Supplementary Fig. 2b). Given that sufficient light intensity is essential for effective phototherapy, a top-emitting NIR OLED structure was specifically designed to ensure photon delivery independent of the inherent limitations of polyester. The detailed designs and optical considerations for this approach are discussed in the following section.
To provide additional protection, an identical nanolaminate TFE layer (30 nm) was deposited via ALD on the top of OLED structure. Finally, a 500 nm parylene-C layer was applied via CVD as an outermost encapsulation layer to prevent direct skin contact, ensuring enhanced durability and user safety (Fig. 1c).
The top-emitting OLED architecture introduces a significant refractive index contrast at the device-to-air interface, resulting in considerable optical confinement due to total internal reflection. This hybrid encapsulation, consisting of the upper TFE and outermost parylene-C layer, not only provides needed mechanical protection but also enhances light extraction efficiency (Fig. 3). Details on the light-extraction enhancement by the hybrid capping are provided in a subsequent section.
The final device architecture was confirmed using scanning electron microscopy (SEM) and cryo-focused ion beam (cryo-FIB) cross-sectional imaging. Milling at room temperature poses a risk of damaging the polyester-based textile substrate due to its low thermal tolerance, potentially leading to mechanical distortion. Hence, cryo-FIB SEM was employed to minimize thermal effects while enabling precise structural analysis. This method facilitated high-resolution imaging, clearly resolving the textile substrate, adhesive and planarization layers, and parylene-C encapsulation of all OLED components without compromising structural integrity (Fig. 1d).
To evaluate the practical applicability of the textile-based NIR OLEDs in wearable phototherapy systems, a prototype NIR OLED-based wearable device was implemented and its integration into various commercially available hat forms was demonstrated. The textile OLEDs were embedded onto representative hat platforms, including bucket hats and baseball caps, to show flexibility and operational reliability under realistic usage conditions.
Operation of the OLED array on a commercial hat/cap platform, while maintaining the flexibility required for a wearable device, was demonstrated by mounting the array on the textile and interfacing it with a flexible printed circuit board (FPCB) that provided array driving and section‑wise switching for scalp‑specific operation. The array comprised NIR OLEDs with emission areas of 69 mm² and 1000 mm², each wired to an independent FPCB channel to enable individual on/off control of each scalp section. An image of the textile‑based NIR OLED device integrated into a commercial bucket hat during operation shows stable performance and uniform emission across the fabric surface (Fig. 1e). The integrated devices exhibited stable on/off switching and maintained consistent electroluminescence under repeated bending (Supplementary Movie 1). Furthermore, a photographic comparison of a mannequin head fitted with the textile NIR OLED device in the off and on states illustrates the contrast in NIR emission (Fig. 1f). In both hat types, the textile OLEDs remained operational under realistic wearing conditions (Supplementary Movies 2, 3).
Furthermore, a wireless phototherapy device composed entirely of flexible components was fabricated to validate portable operation. The system comprised a FPCB for switch-based on/off control and battery-driven OLED operation, the NIR OLED, and a flexible battery (Supplementary Fig. 3a, b). The individual component thicknesses were ~90 μm (FPCB), ~230 μm (textile), and ~450 μm (battery), and the maximum local stack thickness within the hat form factor was ~2.0 mm (Supplementary Fig. 3c–f). Switch-controlled on/off operation of the textile-based OLED stripe integrating fully flexible components was demonstrated without the use of a benchtop power supply (Supplementary Movie 4). For a cap-type platform integrating multiple OLED stripes, continuous emission was maintained during donning and doffing (Supplementary Movie 5).
Clinically validated light-therapy systems for hair loss, such as helmet- and headband-type devices, have demonstrated therapeutic effects but rely on rigid platforms that are primarily suited for indoor use. In this study, the textile-based NIR OLED was implemented in a hat-type platform to propose a phototherapy device design that can be used in both indoor and outdoor settings. Compared with conventional rigid systems, this textile-integrated design offers mechanical flexibility and conformal contact with the scalp and employs an OLED-based NIR area source rather than point-like emitters such as red LEDs or lasers, enabling more uniform illumination over the treatment region (Table 1).
Characterization of Top-emitting NIR OLED
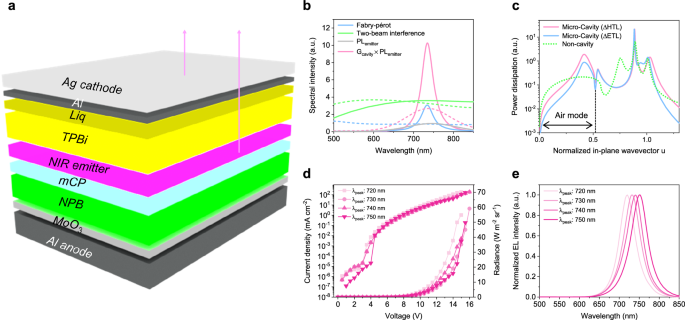
The microcavity-based top-emitting NIR OLEDs are designed to harness optical interference effects for enhanced emission efficiency and spectral control. The device structure comprises a fully reflective anode, serving as a bottom mirror, and a semi-transparent cathode with a reflectance of approx. 70–80% (Fig. 2a). Hence, the light is emitted through the semi-transparent cathode, which is positioned on the opposite side of the textile-based substrate. This configuration induces an interference of light called micro-cavity effect, where light generated within the emissive layer (EML) undergoes multiple reflections between the highly reflective anode and the semi-transparent cathode, modulating the spectral output. The micro-cavity effect is defined as a function of the reflectance and transmittance of each electrode, as well as the distance between the electrodes, referred to as the cavity length.
a Schematic of the top‑emitting NIR OLED employing a microcavity formed by a bottom reflective mirror and a top semi‑transparent electrode. b Microcavity effect divided into Fabry–Pérot resonance and two-beam interference, plotted as relative intensity (y-axis) versus wavelength (x axis), with Ag (solid, strong cavity) and ITO (dashed, weak cavity) as the top electrodes. c Power‑dissipation spectra at λ = 730 nm for top‑emitting OLEDs in which the optical cavity length is controlled by adjusting either the HTL or ETL thickness. d Current density–voltage–radiance (J–V–R) characteristics of the OLEDs for each cavity length. e Electroluminescence spectra showing peak positions tuned from 720 to 750 nm as a function of cavity length.
The micro-cavity effect was examined under varied conditions with ITO (150 nm) and Ag (30 nm) as the top electrode materials (Fig. 2b). This micro-cavity term, derived from Fabry-Pérot resonance and two-beam interference, was calculated using a 2 × 2 transfer matrix simulation28. The results indicate that when the cathode is ITO (dashed line), the micro-cavity term remains relatively constant, exhibiting minimal wavelength-dependent variations. In contrast, the Ag cathode (solid line) significantly enhances the micro-cavity effect within specific wavelength bands, amplifying light emission at the targeted spectral regions.
Figure 2c presents the power dissipation spectra at λ = 730 nm for top-emitting OLEDs, highlighting the effects of different cathode materials and cavity length tuning strategies. The dissipation profiles show that when ITO is employed as the top electrode (non-cavity), the proportion of the air mode is lowered in comparison with cavity OLEDs (i.e., OLEDs with micro-cavity effect) utilizing an Ag cathode. In addition, to investigate which organic layer more effectively enhances the air mode portion, mode distributions were analysed by adjusting the thicknesses of the HTL and ETL, respectively. To compare the two tuning strategies, the optical cavity length was maintained constant by applying an identical thickness increment to either the HTL or ETL. In the HTL‑tuned OLED, the NPB layer was increased by 37 nm relative to the conventional recipe, which adjusted the optical path and positioned the main EL peak at 730 nm. Using this optimized condition as a reference, the ETL‑tuned OLED was fabricated by extending the TPBi layer by the same 37 nm, thereby preserving an equivalent cavity length across both configurations. The microcavity top-emitting OLED, in which the cavity length was tuned via HTL thickness, exhibited an air mode proportion of 38.9%. In contrast, tuning via ETL thickness yielded an air mode proportion of 24.2%, while the non-cavity OLED with an ITO top electrode exhibited an air mode proportion of 18.4%. Such adjustment effectively suppressed the surface plasmon polariton (SPP) mode, thereby enhancing light extraction through the air mode. This improvement shows that the photon extraction can be facilitated by increasing the air mode ratio of cavity structure, thereby improving overall light outcoupling efficiency. In fact, in cavity OLEDs, cavity length modulation through HTL thickness resulted in a maximum external quantum efficiency (EQEmax) of 7.8%, whereas tuning cavity length via ETL thickness yielded a EQEmax of 3.6%. This indicates that HTL-tuning led to approximately a 2.1-fold increase in EQEmax compared to ETL-based tuning strategy (Supplementary Fig. 4a). The observed enhancement is attributed not only to the increased air-mode ratio but also to better carrier transportation arising from layer thickness adjustments. Because hole transport in NPB exceeds electron transport in TPBi, adjustment of the NPB thickness, in part owing to the carrier mobility imbalance29, positioned the recombination zone more effectively within the emissive layer, to reduce the proportion of non-radiative interfacial exciplex. In fact, the HTL-tuned OLED exhibited slightly improved operational lifetime compared with the ETL-tuned device (Supplementary Fig. 4b).
As shown in Supplementary Fig. 5, there existed a red shifting as the cavity length increased. More specifically, a 10 nm variation in cavity length corresponded to a 15 nm shift in the EL spectral peak. This result demonstrates that the optical characteristics of the NIR OLED, especially the peak position, can be tuned through microcavity modulation without modifying the emitter. The recorded max. radiances varied with wavelength, ranging from approximately 40 to 60 W m⁻² sr⁻¹, validating the tunability and high radiance (Fig. 2d). EL spectrum shown in Fig. 2e illustrate tunable emission band over the 720–750 nm region with NIR emitter [Pt(fprpz)2] that underwent spontaneous self-assembly during thin film formation30,31, allowing fine spectral tuning in 10 nm increment. Furthermore, NIR band centered at ∼800 nm can be also fabricated by increasing the cavity length (Supplementary Fig. 6a). The device exhibited low leakage current levels and radiance exceeding 10 W m⁻² sr⁻¹ (Supplementary Fig. 6b).
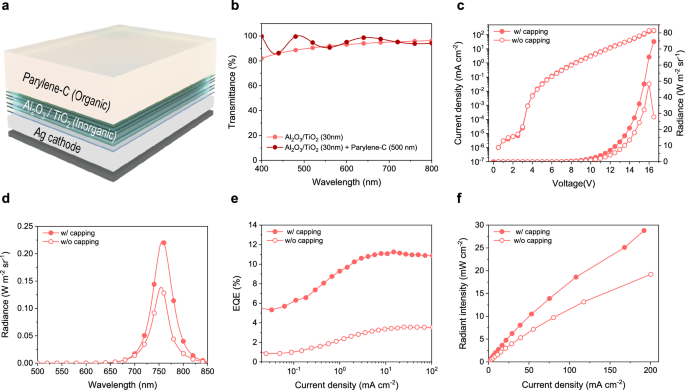
In top-emitting OLEDs, a capping layer is required to facilitate light extraction, owing to the large refractive index mismatch between air (nair = 1) and the emitting medium (nemitter = 1.96), which causes internal light trapping. A capping layer was designed to enhance out-coupling efficiency and was subsequently incorporated into the device architecture (Fig. 3a). It not only improves OLED efficiency but also ensures good device flexibility and operational reliability through a hybrid structure comprising inorganic and organic materials. The inorganic layer, consisting of Al2O3/TiO2 nanolaminate, protects the device from both moisture and oxygen, while the parylene-C organic layer enhances the biocompatibility, which is particularly crucial for applications involving direct contact with human skin.
a Structure of the emitting side showing a hybrid encapsulation stack composed of an inorganic Al2O3/TiO2 nanolaminate and an organic parylene‑C layer, which together function as encapsulation and as an optical capping layer. b Optical transmittance of the capping layers, comparing an inorganic stack and an inorganic–organic hybrid on a glass baseline. c J–V–R characteristics for devices with and without the capping layer (closed symbols, with capping; open symbols, without capping). d Electroluminescence spectra measured at a current density of 21 mA cm⁻². e, f External quantum efficiency and power density as functions of current density, showing the effect of the hybrid capping layer on device performance.
The refractive index of the Al2O3/TiO2 nanolaminate is 1.87, and that of parylene-C is 1.6 in the NIR region. The thickness of the organic layer was optimized to maximize light outcoupling from the OLED, revealing that 500 nm resulted in the most efficient light extraction (Supplementary Fig. 7). By depositing this capping layer over the cathode, reduced the fraction of light trapped by total internal reflection, thereby facilitating more efficient photon extraction. Furthermore, the capping layer enhanced the Fabry-Pérot contribution in the NIR spectral region, leading to an increased NIR emission intensity (Supplementary Fig. 8). Next, the transmittance was measured for both the nanolaminate inorganic layer (30 nm) and hybrid structure comprising the same inorganic layer and an additional 500 nm-thick organic parylene-C film (Fig. 3b). A glass substrate was used as the baseline reference. The nanolaminate inorganic layer exhibited a transmittance exceeding 95% in the NIR region (700–800 nm). Even after the deposition of the organic parylene-C layer, transmittance was maintained over 90% transmittance in the NIR region.
To investigate the influence of the capping layer on device performance, the optoelectronic characteristics of OLEDs were compared with and without the added layer through 10 devices. Integration of the capping layer did not alter the electrical characteristics, as evidenced by unchanged input bias characteristics, whereas an enhancement in radiance was observed (Fig. 3c). After encapsulation, the maximum radiance increased from 44.8 W m⁻² sr⁻¹ to 78.7 W m⁻² sr⁻¹, accompanied by a 3 nm redshift of the main peak and a 7 nm broadening of the FWHM (Supplementary Fig. 9a). Under identical current-driving conditions, a radiance of 12.3 W m⁻² sr⁻¹ was measured for the device with the capping structure, compared with 7.4 W m⁻² sr⁻¹ for the uncapped counterpart (Fig. 3d). This improvement is attributed to an enhanced light outcoupling efficiency, likely facilitated by the modified optical interference condition introduced by the capping structure. Furthermore, the functional contribution of the capping layer was validated through EQE and power density characteristics. The maximum EQE increased from 3.7% at a current density of 30 mA cm⁻² without the capping layer to 11.4% under the same current density with the capping layer. In addition, at a higher current density of 100 mA cm⁻², an EQE of 10.8% was achieved (Fig. 3e). Similar trend was observed in devices without encapsulation, indicating that the low level of roll-off, as shown in Supplementary Fig. 9b. The maximum power density, calculated from the angular distribution of each device, increased from 19 mW cm⁻² to 33 mW cm⁻² (Fig. 3f).
Stability of wearable NIR OLEDs
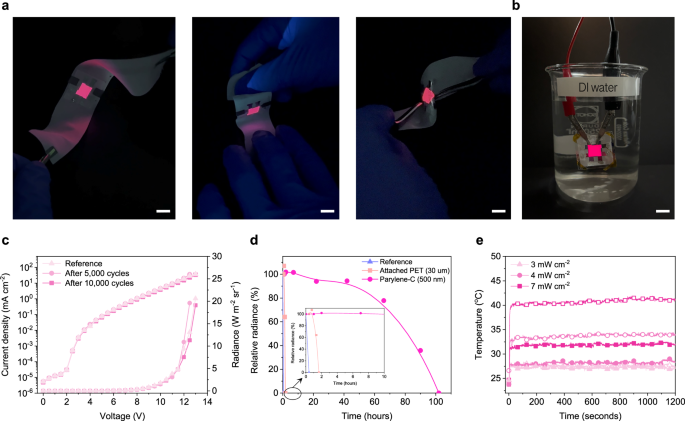
The mechanical and deformational stability of textile-based NIR OLEDs was evaluated to establish their feasibility for wearable applications. To implement the developed NIR OLED structure on a wearable configuration, only the substrate material was changed, while the fabrication process remained identical. Despite the change in substrate, the corresponding EL characteristic of NIR OLED was preserved, as shown in Supplementary Fig. 10a. In addition, device stability was maintained without any detectable leakage paths, and the emitted light intensity exceed 10 W m-2 sr-1 at approximately 12 V and a current density of 31.5 mA cm-2 for the textile device, the minimum level required for phototherapy (Supplementary Fig. 10b)7. Continuous operation was demonstrated under bending, wrinkling, and submersion owing to the inherent flexibility of the NIR OLEDs and the textile platform, without immediate degradation observed (Fig. 4a, b).
a, b Photographs of the textile‑based NIR OLED during operation under mechanical deformation (scale bar, 10 mm): a bending and wrinkling; b immersion in deionized water. c J–V–R characteristics after 5,000 and 10,000 bending cycles at a tensile strain of 2.1% (bending radius, 2 mm). d Relative radiance as a function of immersion time in deionized water for devices with no encapsulation, with attachable polyethylene terephthalate (PET), and with parylene‑C encapsulation. e Surface temperature of the device during continuous operation for 20 min; open symbols denote devices without a thin heat‑sink layer, and closed symbols denote devices with a heat‑sink layer.
Flexibility of the textile-based device was assessed by cyclic bending at a radius of 2 mm, corresponding to an applied tensile strain of 2.1%. Finite element analysis indicated that, for the textile platform with a woven warp and weft architecture, this condition represented the maximum point at which the applied strain could be defined24. The J–V–R characteristics remained stable after 5000 and 10,000 bending cycles, with no evidence of electrical leakage, mechanical delamination, or degradation in radiance output (Fig. 4c), indicating robust structural reliability.
Submersion stability of OLEDs was assessed by evaluating their water resistance through immersion testing. Various encapsulation strategies were systematically investigated for their ability to preserve device integrity under submerged conditions (Fig. 4d). Devices encapsulated solely with an inorganic barrier layer showed significant degradation within 10 min of immersion. In contrast, OLEDs protected with a 30 μm-thick PET layer applied retained operational stability for up to 1 h before deterioration was observed. Notably, OLEDs encapsulated with a 500 nm-thick parylene-C layer developed in this study exhibited water resistance, maintaining over 90% of their initial radiance after 50 h of continuous submersion. Performance degradation became pronounced only after prolonged exposure, with radiance falling below 50% following 80 h of immersion.
To validate the risk of skin burns upon contact, the temperature of the device was monitored. This was analysed for a continuous operation under various radiation levels during 20 minutes (Fig. 4e). Regardless of applied current density, their temperature levelled shortly after initiation. The devices reached 28.0 °C at 3 mW cm⁻², 34.0 °C at 4 mW cm⁻², and 41.0 °C at 7 mW cm⁻², respectively. The polyester-based textile platform used in this study possesses a relatively low thermal conductivity (0.3–0.4 W/m·K), significantly lower than that of glass ( ~ 1 W/m·K). Such thermal accumulation leads to heat retention in the active layer during prolonged operation, thereby accelerating device degradation. Reducing heat accumulation and suppress OLED degradation caused by heat, a heat sink tape was attached to the textile devices. As a result, their temperatures were further reduced to 27.5 °C, 28.0 °C, and 32.0 °C, ensuring safe operation against possible skin burns. Additionally, to assess long-term device reliability, operational lifetime was evaluated on different substrates. OLEDs exhibited comparable LT50% performance regardless of whether glass or textile substrates were used (Supplementary Fig. 11a, b). Furthermore, the incorporation of a heat sink extended the lifetime for both configurations. Notably, the enhancement was more pronounced in textile-based OLEDs. Devices with a heat sink demonstrated up to a two-fold increase in LT50% compared to those without it. This improvement is attributed to the effective dissipation of heat generated during operation, which reduces thermal degradation on the OLED stack.
Penetration of NIR irradiation
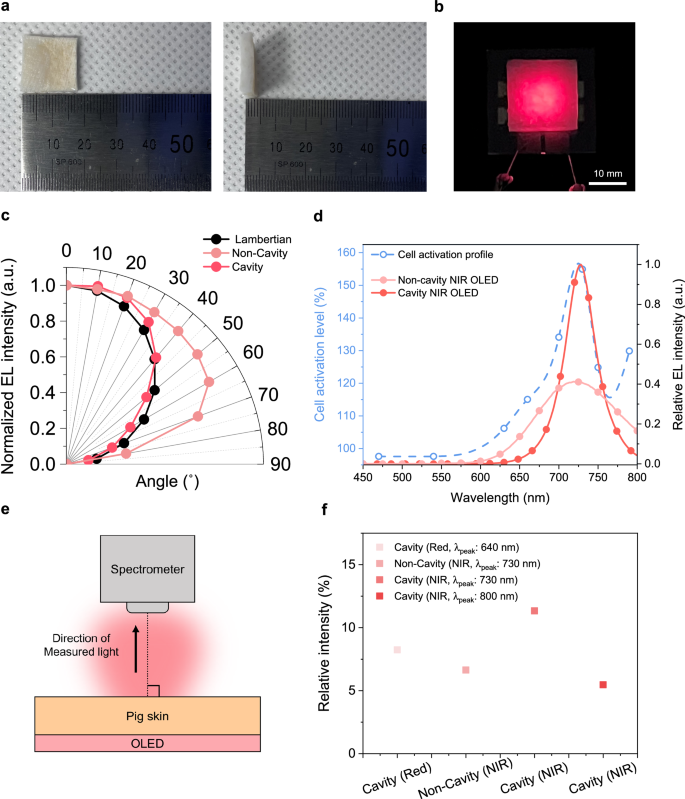
Non-invasive phototherapy requires sufficient light penetration into the skin to stimulate biological targets without surgical intervention. Since human hair follicles are typically located approximately 3 mm beneath the skin surface, achieving light transmission to this depth is critical for effective light delivery to dermal papilla cells within the follicle niche. To assess whether the NIR OLED devices meet this requirement, pigskin was employed as a tissue model due to its structural and optical resemblance to human skin, enabling analysis of in vivo light propagation.
As shown in Fig. 5a, the pigskin sample is shown together with its front view and thickness. The experimental setup used to evaluate light transmission through pig skin, with the OLED positioned behind the sample, is shown in Fig. 5b. To enable accurate measurement of transmitted light intensity, the OLED emitting area was fully covered by the skin sample. Because OLEDs exhibit a broad angular emission unlike narrow beam lasers or LEDs and scattered light arising from melanin and other tissue components in pig skin also needed to be captured. Consequently, the pigskin area (324 mm2; 18 mm × 18 mm) was set to approximately five times the OLED emitting area (69 mm2; 8.3 mm × 8.3 mm), thereby ensuring full coverage for reliable measurement.
a Photographs of the porcine skin sample (18 × 18 mm², thickness 3 mm) showing front and side views. b Experimental configuration with the epidermal surface positioned in front of an OLED emitting surface. c Angular emission distributions of cavity and non‑cavity NIR OLEDs. d Overlap between the action spectrum of human dermal papilla cell for proliferation15 and the electroluminescence spectra of the cavity and non‑cavity NIR OLEDs at equal radiance. e Schematic of the setup for normal‑incidence transmission measurements. f Relative transmitted intensity for different OLED emissions through porcine skin at constant radiance.
To assess the effects of OLED emission wavelength and angular distribution for non-invasive phototherapy, transdermal transmission was analyzed using four OLED device types, including a cavity red OLED with a peak at 640 nm, a non-cavity and a cavity NIR OLED with peak at around 730-740 nm, and a cavity NIR OLED with peak at 800 nm (Supplementary Fig. 12). Angular emission profiles of the cavity and non-cavity NIR OLEDs were compared to a Lambertian reference distribution (Fig. 5c). The non-cavity OLED exhibited a super-Lambertian emission pattern, while the cavity OLED showed an angular profile closely matching that of a Lambertian emitter. Although cavity OLEDs typically demonstrate sub-Lambertian behavior due to their optical interference effects, the cavity OLED developed in this study exhibited near-Lambertian characteristics. This deviation is attributed to prior optimization of the ETL thickness in the non-cavity NIR OLED structure30,32, which may have increased lateral scattering toward oblique angles (i.e., viewing angles exceeding 10°). In contrast, the cavity structure enhanced forward emission, achieving an angular distribution closer to a Lambertian profile. A blue shift of the EL spectrum with increasing viewing angle was observed in cavity NIR OLEDs, attributable to microcavity resonance that shifts the cavity mode toward shorter wavelengths with increased viewing angles (Supplementary Fig. 13).
The cavity red and the cavity NIR (800 nm) OLEDs exhibited sub-Lambertian and super-Lambertian angular distributions, respectively, as shown in Supplementary Fig. 14. The 800 nm device, which was red-shifted by approximately 60 nm from the emitter’s photoluminescence peak (730–740 nm) due to microcavity effect with increased optical path, exhibited increasing intensity at oblique angles compared to the normal direction. This super-Lambertian profile shows that when the spectral peak shifts significantly from the intrinsic PL peak, a relatively higher portion of emitted light is directed toward off-normal angles compared to the forward direction.
The EL spectra obtained under equal radiance revealed a narrowing of the spectral bandwidth upon cavity integration. The FWHM decreased from 130 nm in the non-cavity device to 45 nm in the cavity OLED. At the same time, emission intensity in the 730–740 nm region was enhanced by the cavity configuration. At normal emission, the band‑integrated photon radiance within the region was approximately 2.4 times greater for the cavity NIR OLED than for the non‑cavity device. This tuning can provide to greater photon delivery at the hDPCs activation peak near 730 nm, compared with the non-cavity device, thereby enhancing photostimulation under equivalent dose condition, as shown in Fig. 5d. An analysis relating hDPCs activation to the OLEDs’ optical properties is provided in the in vitro section.
Transdermal light propagation under realistic operation was assessed by placing the emitting surface of each OLED in direct contact with epidermis of pig skin (Fig. 5e). Transmittance was defined as the ratio of the radiance measured at the dermal exit plane along the normal direction to the radiant output of the OLED. As shown in Fig. 5f, the cavity NIR OLED achieved the highest transdermal transmission efficiency at 12%, surpassing the cavity red OLED (8%), the non-cavity NIR OLED (6%) and the NIR OLED with an 800 nm peak (5%). The transmission of the cavity NIR OLED shows the improved tissue penetrability of longer wavelength NIR emissions (700–800 nm) compared to shorter wavelength red emissions (600–700 nm). Furthermore, the enhanced transmission observed in the cavity NIR OLED compared to its non-cavity counterpart suggests the role of forward-directed emission for non-invasive light therapy.
To validate transmittance of the NIR OLED light in real skin, Monte Carlo simulations were performed to measure photon delivery from a skin conformal wearable near infrared OLED to the hair follicle region. In this model, the epidermis, dermis, and subcutaneous fat layers were defined with layer-specific refractive indices scattering coefficients (µs), and anisotropy factors (g), all set as wavelength-dependent values from the literature33. The emission source was set an 8.3 × 8.3 mm² square that was placed in direct contact with the epidermal surface (Supplementary Fig.15a). Planar receivers were positioned normal to the surface at depths from 1.0 mm to 5.0 mm. The ratio of receiving surface irradiance to power density of emitting surface was 4.3% at 3.0 mm, 1.4% at 4.0 mm, and 0.5% at 5.0 mm (Supplementary Fig.15b).
Simulated transmission was lower than the measured value in porcine skin. The difference was attributed to two features of the experimental setup. The skin was dried before measurement, which reduced absorption by water. In addition, the samples consisted only of epidermis and dermis, whereas the model included epidermis, dermis, and subcutaneous fat. However, the receiving ratio at depths of 3 to 4 mm was similar to that in our in vitro setup, which showed a PBM effect under OLED irradiation as described in the below section.
In vitro PBM induced by OLED irradiation
To validate the phototherapy effects of NIR OLEDs on hDPCs and their potential for hair regeneration applications, this study conducts an in-vitro tests. HDPCs are specialized mesenchymal cells situated within the dermal papilla at the base of hair follicles. They play a central role in hair follicle regulation by secreting critical growth factors, including fibroblast growth factor-7 (FGF-7) and insulin-like growth factor-1 (IGF-1), which directly influence follicular growth and cycling34,35,36,37.
Promoting the migration of hDPCs is crucial for effective cell-cell communication and follicular regeneration, particularly during an anagen phase. Moreover, preventing or delaying cellular senescence—an irreversible state of growth arrest—is vital for maintaining follicular health and mitigating age-related hair loss37. Enhanced migratory capacity and anti-aging properties of hDPCs represent significant factors for hair loss therapy.
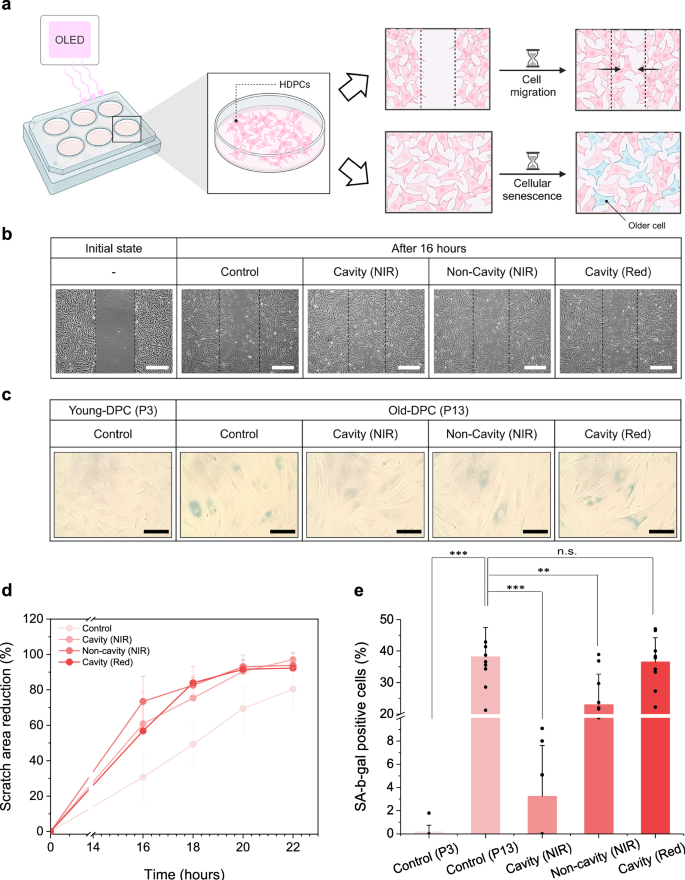
Effects of light exposure on cell migration and cellular senescence of hDPCs were examined. To validate the influence of light treatment on biological responses, a cell migration assay and a cellular senescence assay were performed. For the migration assay, a scratch was made at the center of a cell culture to create an empty space. NIR emission was then irradiated onto the scratched region, and the extent of cell movement into the gap was observed over time to evaluate the migration. For the senescence assay, senescent cells were stained with blue color using a standard β-galactosidase staining method, and changes in the number of stained cells were monitored following NIR OLED irradiation to investigate its potential to suppress cellular aging (Fig. 6a).
a Schematic of the in vitro assay in which cultured human dermal papilla cells (hDPCs) in a six‑well plate were irradiated by the NIR OLED. Cellular responses were assessed by cell migration and cellular senescence. Migration was quantified from the closure of scratched areas over time, and senescence was evaluated by β‑galactosidase (SA‑β‑gal) staining. b Optical micrographs acquired 16 h after irradiation showing migration into the scratch region (scale bar, 500 μm). c Micrographs acquired after light exposure for senescence analysis (scale bar, 300 μm). d Relative scratched area quantified from 16 to 22 h at 2 h intervals. Data are shown as mean ± s.d. (n = 6 per group and time point), where each data corresponds to the closure rate in a single non-overlapping microscopic field of view. e Fraction of SA-β-gal-positive cells. Data are shown as mean ± s.d. (n = 10 per group). Each data corresponds to the fraction of SA-β-gal-positive cells in a single non-overlapping microscopic field of view. P values were obtained from a two-sided unpaired Student’s t-test without adjustment for multiple comparisons; **p < 0.01, ***p < 0.001.
Three OLED types were employed to assess the effect of optical properties on cell activation, including the cavity NIR OLED fabricated in this study and two devices used in the transdermal section, a cavity red OLED and a non-cavity NIR OLED. Device characteristics including peak wavelength and full width at half maximum were summarized in Table 2.
The OLEDs were positioned 15 mm above the well plate to mitigate thermal effects on cells (Supplementary Fig. 16a). For each OLED, the emitting‑surface radiance was set to around 10 W m-2 sr-1 for continuous irradiation during 20 min. Analysis of cell activation was limited to an orthogonally aligned square region, where the emitting surface measured 8.3 × 8.3 mm² and the area matched the beam size of OLED (Supplementary Fig. 16b).
To verify the light dose delivered to the cell medium, a Monte Carlo simulation that reproduced the in vitro configuration was performed. Because dose delivery to away from emitting surface depends on the OLED angular emission profile, the receiving surface was defined as the full well with a diameter of 35 mm. The simulation indicated an energy spread for the non-cavity NIR OLED, with an effective illuminated diameter of approximately 6.0 mm as shown in Supplementary Fig. 16c. The cavity NIR OLED exhibited an effective illuminated diameter of approximately 4.5 mm (Supplementary Fig. 16d). This observation is consistent with the more diffuse super Lambertian emission of the non-cavity device. However, to ensure that in vitro outcomes were obtained from cells exposed to light, the analysis area was fixed to match the beam size and aligned orthogonally to the OLED.
Under these conditions the effective irradiance at the cell medium was 0.06 mW cm⁻², which is approximately 2% of the emitting surface power density of 3 mW cm⁻², and this corresponded to a delivered irradiance of 0.072 J cm⁻² during 20 min of continuous exposure. This configuration allowed to compare cellular responses under same dose to red versus NIR emission and to evaluate the impact of NIR spectral bandwidth.
Cell activation after OLED exposure was examined by optical microscopy of hDPC cultures. For the migration assay, optical microscopy (OM) images were acquired immediately after scratch formation and 16 h after OLED irradiation, and greater closure of the scratched area was observed in both red- and NIR-irradiated groups compared with the non-irradiated control (Fig. 6b). Cellular senescence was evaluated by SA-β-gal staining at two passages. Passage-3 and passage-13 cells were used as references. The experimental group was composed of passage-13 hDPCs. Based on SA-β-gal staining, the lowest fraction of positive cells was observed in passage-3 hDPCs despite the absence of light exposure, consistent with their younger passage. Across all experimental groups, the proportion of stained cells decreased relative to the control. However, a greater reduction was achieved with NIR irradiation than with red, and the cavity-tuned NIR OLED was associated with the most pronounced decrease (Fig. 6c).
Cell migration during the 16 to 22 h period after OLED irradiation was quantified as the filled‑area ratio relative to the initial scratch area (Fig. 6d). Interval observations at 2 h increments from 16 to 22 h were performed, and progressive filling of the scratched region by migrated cells was observed (Supplementary Fig. 17). At 16 h, the control filled 30.7% of the scratched area, whereas closure reached 60.9% in the cavity NIR OLED group, 73.5% in the non‑cavity NIR OLED group, and 56.8% in the red OLED group. Over time, the gap continued to close and the migration rate reached saturation. At 22 h, closure reached 97.1% in the cavity NIR OLED group, 93.8% in the non‑cavity NIR OLED group, and 92.3% in the red OLED group, exceeding the 80.5% observed in the control (Table 2).
Cellular senescence was defined as the proportion of SA-β-gal-positive cells and was calculated by dividing the number of stained cells by the total cell count. Results are presented in Fig. 6e and Table 3. With identical culture duration, SA-β-gal positivity in passage-3 hDPCs was 0.18%, and few blue-stained cells were observed in OM images (Supplementary Fig. 18a). Without light exposure, SA-β-gal positivity in passage-13 hDPCs was 38.2% (Supplementary Fig. 18b). The difference between untreated passage-3 and passage-13 cultures is attributable to passage effects and is consistent with lower baseline senescence at younger passages. Under cavity red OLED irradiation, SA-β-gal positivity was 36.5% (Supplementary Fig. 18c). Under NIR exposure, SA-β-gal positivity was lower than under red exposure, at 3.2% in the cavity-tuned NIR group and 23% in the non-cavity NIR group (Supplementary Fig. 18d, e). These findings are consistent with the hDPCs activation profile in Fig. 5d. Compared with red exposure, lower senescence fractions were obtained under NIR exposure, with the lowest value observed for the cavity-tuned NIR OLED, which is characterized by higher photon amount in the 730–740 nm band.
Discussion
In summary, a textile integrated NIR OLED is presented as a light source for non-invasive wearable phototherapy for hair loss. The study reports development of customized NIR OLED architecture for PBM with microcavity tuning near 730 to 740 nm, integration of the device into a portable wearable hat prototype, and validation of PBM efficacy for hair loss under NIR OLED illumination.
Through microcavity tuning, the emission spectrum of the NIR OLED can be adjusted to 720–800 nm and power density is reached above 3 mW cm⁻² (around 10 W m⁻² sr⁻¹ radiance) that is minimum intensity to induce PBM. Application‑specific spectra can be achieved by aligning the cavity mode with cellular action band.
The NIR OLEDs were integrated onto a polyester-based textile platform. The device’s J–V–R and EL performance was preserved after bending to a 2 mm radius for 10,000 cycles, owing to the flexibility of the NIR OLED structure and the textile platform. Despite increasing interest in NIR emitters17, flexible NIR OLEDs with comparable flexibility have been reported yet.
Relative to red light commonly used in conventional phototherapy devices, a higher subsurface irradiance can be delivered to the dermal papilla during non-invasive treatment with NIR OLEDs. This is attributed to the reduced scattering and absorption of NIR light by skin tissues. In addition, unlike LEDs and lasers, OLEDs allow broader and more freely control of the emitting surface, thereby enabling deeper penetration. Given that light penetration is promoted when the beam diameter is at least ~10 mm19, OLEDs are more advantageous than point light sources.
A key advantage of the NIR OLEDs for PBM application is spectral tunability. The emission characteristics of the NIR device can be customized via the microcavity effect to enhance photon flux in the 730–740 nm band, aligning with the activation spectrum of hDPCs. According to the in vitro tests, all experimental groups exposed to OLED irradiation were observed to show increased hDPC migration and reduced cellular senescence relative to untreated controls. However, the level of cell activation differed with the optical characteristics of the OLED. The anti-senescence effect under NIR was greater than under red illumination, and the customized NIR device outperformed the non-customized variant, yielding ~91.6% improvement relative to the control. These results indicate that microcavity-enabled spectral alignment and concentrated photon delivery within the target band are key determinants of PBM performance.
The cellular response to PBM is highly dependent on the irradiation wavelength, with maximal bioactivity observed at or near the absorption maxima of cytochrome c oxidase (CCO)35. When cells were irradiated with wearable NIR OLEDs precisely tuned to these CCO absorption peaks—while maintaining identical light dosages—significantly enhanced cellular activation was observed15. This enhancement is attributed to selective photoactivation of CCO, which initiates a mild increase in mitochondrial reactive oxygen species (ROS) production38. The controlled ROS elevation subsequently induces a hormetic stress response, upregulating antioxidant defenses and DNA repair mechanisms39. Such ROS-mediated hormesis effectively delays the onset of cellular senescence.
The textile‑integrated NIR OLED system offers advantages as a biocompatible platform for non‑invasive phototherapy. The microcavity structure enables precise spectral control within the therapeutic range, effectively matching the activation spectrum of dermal papilla cells. Using parylene‑C, which is FDA‑approved material, as both a planarization and encapsulation layer for textile‑based OLEDs ensures biocompatibility at the skin interface24,26. This material selection strategy enables safe, non‑invasive wearable phototherapy in close contact with the skin. In addition, textile‑based OLEDs, composed entirely of flexible materials including textile substrate and OLED light source, could be close to the scalp surface and provide uniform illumination of targeted regions with minimal irradiance variation. Furthermore, we have demonstrated a prototype in which embedding the device in a hat‑type form factor facilitates natural wearability and comfort, allowing users to receive phototherapy during routine daily activities without the need for benchtop instrumentation, showing its potential as a wearable device.
Despite these advances, several constraints for translation remain. Microcavity OLEDs are characterized by view‑angle–dependent spectral shifts, which reduce overlap with cellular action spectra at oblique incidence. Furthermore, portable operation and device flexibility are limited by trade‑offs among deliverable energy density, total device thickness, and capacity of the power‑supply components required for OLED driving.
In addition, hair-loss pathology involves multiscale interactions that extend beyond hDPC responses, including crosstalk with epithelial stem cells, immune system, the vasculature and systemic endocrine signals. Therefore, the results obtained from our in vitro tests should be interpreted as evidence of photobiological modulation at the cellular level rather than as direct proof of hair-loss treatment. To establish whether wavelength-optimized textile-integrated NIR OLEDs can ultimately provide clinically meaningful benefits, future studies will need to include controlled in vivo models and, eventually, clinical trials to evaluate changes in hair density, hair-shaft thickness and hair length.
To reach real-world applicability, further technological refinement is required. The development of flexible batteries with sufficient voltage and power capacity is essential for commercial deployment. Reducing overall thickness through redistribution of the FPCB and battery across alternative hat regions would improve user comfort and decrease localized bulk. In addition, practical use demands that washability and mechanical flexibility be achieved simultaneously. Attaining washability requires comprehensive waterproofing at the device level and reliable sealing of interfaces among the battery, FPCB, and OLED. Although full washability has not yet been implemented in the current prototype, component-level studies on waterproof OLED encapsulation40,41, flexible batteries42,43, and circuit boards are ongoing. These efforts are expected to fabricate a fully washable and stable system capable of sustained operation in daily use44, thereby advancing the textile-integrated NIR OLED platform toward practical, commercially viable wearable phototherapy.
Methods
NIR OLEDs fabrication
NIR OLEDs were fabricated by thermal evaporation (base pressure, 2 × 10⁻⁶ Torr). Organic and electrode layers were sequentially deposited on the substrate under this vacuum. The Al anode (100 nm) and Ag cathode (30 nm) were deposited at 2 Å s⁻¹. A Pt(II) complex–based NIR emitter was used as the emissive layer30(EML, 20 nm) and was deposited at 0.2 Å s⁻¹. The carrier‑blocking layer, N,N′‑dicarbazolyl‑3,5‑benzene (mCP, 15 nm), the hole‑transport layer, N,N′‑di(1‑naphthyl)‑N,N′‑diphenyl‑(1,1′‑biphenyl)‑4,4′‑diamine (NPB, 87 nm), and the electron‑transport layer, 1,3,5‑tris(N‑phenylbenzimidazole‑2‑yl)benzene (TPBi, 60 nm) were deposited at rates of 0.5–1 Å s⁻¹. The NPB thickness (87 nm) was selected to tune the optical microcavity to a peak emission near 730 nm. The hole‑injection layer, molybdenum(VI) oxide (MoO₃, 10 nm), and the electron‑injection layer, 8‑quinolinolato lithium (Liq, 2 nm), were deposited at 0.5 Å s⁻¹ and 0.1 Å s⁻¹, respectively.
Encapsulation was formed with an inorganic/organic nanolaminate. An Al₂O₃/TiO₂ inorganic layer (30 nm total) was deposited by thermal atomic layer deposition (ALD) at 40 °C using trimethylaluminum (TMA) and tetrakis(dimethylamino)titanium (TDMAT) as metal precursors. An organic parylene‑C layer (500 nm) was subsequently deposited by chemical vapor deposition.
Textile-based NIR OLED fabrication
The textile platform was prepared according to previous research26. Prior to OLED deposition, a 30 nm Al2O3/TiO2 nanolaminate barrier layer was deposited on the textile by ALD. NIR OLED was subsequently fabricated on the textile platform using the same fabrication sequence and recipe described NIR OLED fabrication section.
OLED characterization
OLEDs were driven under constant bias using a Keithley 2400 benchtop source meter. Electrical operating conditions were controlled and logged via computer. Optical measurements were performed using a CS-2000 spectroradiometer (Konica Minolta) and a Blue-Wave spectrometer (StellarNet). Angular emission data were acquired by rotating the light source about the spectrometer normal in fixed increments. Flexibility was assessed using a bending tester from Sciencetown Inc.
Hat-type prototype
To demonstrate operation of the OLED array on a commercial hat/cap platform, the array was mounted on the textile and interfaced to a flexible printed circuit board (FPCB) that provided array driving and section‑wise switching for scalp‑specific operation. The array comprised OLED emitters with emission areas of 69 mm2 and 1,000 mm2, each wired to an independent FPCB channel for individual on/off control. For array‑level characterization, the source was operated in constant‑current mode using a benchtop sourcemeter (Keithley 2636B). The operation is shown in Supplementary Movies 1–3.
A wireless prototype was assembled to evaluate portability. The architecture integrated a flexible battery for portable operation, a flexible printed circuit board providing on/off control, battery power delivery, and OLED interconnection, and the NIR OLED. A 3 V flexible Renata thin-film battery (CP042350) was employed, and 3 or 4 cells were connected in series to achieve the required minimum level of light intensity for phototherapy. All components were integrated on the polyester-based textile to form a single portable module, and operation was performed from the flexible battery without benchtop supply (Supplementary Movies 4, 5).
Pig-skin transdermal propagation
Porcine skin membranes comprising the epidermis and dermis (micropig Franz Cell Membrane, APURES) were used. Membranes were thawed at room temperature for approximately 1 h before use. Each membrane was placed on an OLED with the epidermal surface facing the emission area to measure transdermal propagation. Red OLEDs were fabricated following the protocol described in previous research45. Non-cavity NIR OLEDs were fabricated according to the procedure reported in previous research15. The OLEDs were operated under each condition to achieve same power density, and light transmitted through the skin was measured on the opposite side.
Monte Carlo simulation
Monte Carlo simulations were performed using LightToolsTM. For the in vitro configuration, the geometry of each well and each OLED was reproduced to match the experimental setup. The angular emission profile and the emitting area of each OLED were defined from measured data. For the real skin model, literature-reported optical parameters were used to construct a multilayer skin geometry comprising the epidermis, dermis, and subcutaneous tissue33,46, and scattering was represented by the Henyey–Greenstein scattering phase function. To mimic non-invasive phototherapy, the emitting surface of the OLED was placed in direct contact with the epidermal surface in the model.
In vitro setup
During all irradiations, the distance from the OLED emitting surface to the cell culture plate was set to ~15 mm. A transparent plate lid was placed over the wells to minimize particulate contamination. Each well was filled with cell culture medium such that the vertical optical path length from the inner surface of the lid to the medium surface was ~15 mm. The well plate was placed within the OLED driving equipment, which shielded the wells from ambient light so that only the OLED emission reached the medium. A 6-well plate was used, with one OLED assigned to each well, and cell activation was validated in each well.
Cell culture
hDPCs at passage 5 to passage 13 were obtained (c-12071; PromoCell) and cultured in Follicle DPC Growth Medium (c-26501; PromoCell) with 0.1% Gibco™ antibiotic-antimycotic (Thermo Fisher Scientific, Waltham, MA, USA). Cells were maintained in a humidified incubator at 37 °C under 5% CO2.
Cell migration assay
hDPCs (passage 3) were seeded in a 6-well plate at a concentration of 2.5 × 105 cells/well and incubated until reaching 80% confluence. The cell cultures were scratched with a sterile 1000 μL pipette tip. Detached cells were washed away with PBS (21-030-CVR; Corning). Subsequently, 2 mL of DPC growth medium was added. Horizontal reference lines were drawn on the bottom of the plate with an ultrafine tip marker to create a grid for alignment during image acquisition. The plate was then placed in the illumination device, and light irradiation was applied under controlled conditions. Following irradiation, the plate was placed under a microscope aligned using the reference marks. Selected regions of interest were imaged every 2 h from 16 h to 24 h post-scratch using a microscope (Nikon, ECLIPSE Ts2) with a 4 × objective. Scratch area, wound coverage, and average wound width were quantified using the ImageJ plugin Wound Healing Size Tool.
Cellular senescence assay
hDPCs (passage 13) were seeded in a 6-well plate at a concentration of 1 × 105 cells/well and incubated for 24 h. After light exposure, the cells were cultured for 2 days, placed into a new 6-well plate at half density, and cultured for an additional 4 days. SA-β-gal activity was assessed using a histological staining kit according to the manufacturer’s instructions (#9860S, Cell Signaling).
Reporting summary
Further information on research design is available in the Nature Portfolio Reporting Summary linked to this article.