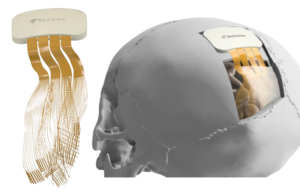
ReVision Implant announced today that it received FDA breakthrough device designation for its Occular visual cortical prosthesis.
Occular interfaces with the brain in an effort to restore functional vision in people with severe blindness. That includes patients whose condition can’t be treated with retinal implants or optic nerve therapies.
According to the company, unlike most experimental vision-restoration technologies, which rely on stimulating the retina, interfacing directly with the brain’s visual cortex enables Occular to bypass damage to the eye or optic nerve while restoring vision.
Occular uses a miniature camera mounted on a wireless headset to capture visual information. It transmits data to a brain implant that stimulates neurons in patterns interpreted by the brain as points of light. These signals combine to form visual representations. They may help users identify objects and navigate their surroundings.
Because the implant stimulates the visual cortex directly, ReVision said it could prove suitable for patients with a wide range of blindness causes. Those include retinal degeneration and optic nerve damage.
The Belgian neurotechnology startup said the breakthrough nod paves the way toward first-in-human clinical trials. It could pit the company against other brain-computer interface (BCI) companies developing vision-restoring implants. That includes Science Corp., which last week raised $230 million to support its platform, and Neuralink, the BCI venture from Elon Musk.
ReVision plans to conduct a first short-term clinical trial during a scheduled brain surgery in October 2026. It then plans to begin early-stage human clinical trials in blind volunteers as soon as summer 2027, subject to regulatory approvals.
“While cochlear implants have transformed treatment for hearing loss, there is still no widely available neuroprosthetic solution for restoring vision,” said Frederik Ceyssens, CEO of ReVision Implant. “Our aim is to provide people living with severe blindness with functional vision that improves independence and quality of life.”