Anything will lase if you hit it hard enough.
-Arthur Schawlow
What do you think of, when you picture a laser? Maybe a laser pointer? Val Kilmer from Real Genius? The Death Star?
Here are some things you probably do not picture:
Peacocks
Mars
Gin and Tonic
Cancer
And yet, all of these things will lase, if you hit them hard enough.
Lase is a serendipitous verb. LASER,1 an acronym for Light Amplification by Stimulated Emission of Radiation, accidentally forms a perfect agent noun and implies a verb; just as the driver drives and the conductor conducts so does the laser lase.2
The dictionary definition gives us:
A laser is a device that emits a highly concentrated, narrow beam of coherent light through a process called Light Amplification by the Stimulated Emission of Radiation. Unlike standard light sources (like light bulbs), lasers produce a single color, with light waves traveling in phase and in a straight line.
This is not fully correct. So then, what does it mean, to lase?
When distilled to its essence, a laser requires three things: the pump, the gain medium, and the cavity. This is a topic close to my heart, born from the hours I spent explaining that an Optical Parametric Oscillator (which I was building) was not a true laser to people who couldn’t care less.

Assuming you, reader, could care less, we need a little background in atomic physics.
Einstein is underappreciated. In 1917, about a year after General Relativity, a decade after Special Relativity, and exactly 100 years before I entered graduate school (coincidence?)3 he published yet another paper that founded an entire discipline: On The Quantum Theory of Radiation.4
I took the opportunity to read it for this post, and it is surprisingly clear (with only two references, one citing himself, lol).
In it, he introduced a completely novel process in atomic systems, which complemented and consummated the two known processes: absorption and spontaneous emission. We now call this stimulated emission. Take a look at the figure below:
Assume we have an electron with two possible states, with energies of 0 (ground) and 𝜖 (excited). Now consider the following three processes.
The electron is initially in the ground state, a photon comes along with exactly 𝜖 energy, it is absorbed and the electron hops up to the excited state.
The electron doesn’t stay excited forever—it eventually decays and in doing so, emits a photon of energy 𝜖. However, this process is not triggered like absorption, it occurs at a random time and emits the photon in a random direction.5
The random direction didn’t bother Einstein so much, because that has a clean analogy to classical physics (isotropic emission on average). But the random timing of the emission bothered him, and was one of the first hints of the irreducible randomness of quantum mechanics, not formalized until four years later.
Einstein’s genius was recognizing that a third process was required in order to explain Planck’s radiation law at a deeper level. An electron in an excited state (not ground!) sees an incoming photon of energy 𝜖, jumps down to the ground state and emits two identical photons. The emission of an extra photon is stimulated by the incoming photon.
This process is an amplifier in a way that no classical process can be. One photon comes in, and two identical photons come out: same frequency, same direction, and quantum mechanically indistinguishable.6
Can you, perhaps, cascade this process? Produce a gigantic mass of identical photons? Burn a hole in the wall with it? Yes you can, and it requires three elements to do it, our gain medium, pump, and cavity.
In our toy examples above, we posited a two-level system, with ground and excited state, but told you nothing about it. In reality, this is the gain medium, an atomic system that has discrete, accessible energy states for its electrons. A lot of the time, media that fluoresce well are also good laser gain media.
The gain medium sets many of the fundamental properties of the laser, most relevant is the set of allowed wavelengths, and so usually, the laser is named after the gain medium.
They generally fall into six categories.7 These categories live mainly in my head, so feel free to quibble with my categorization in the comments. This terminology tends to be what separates the expert from the novice—anyone who’s anyone pronounces Ti:Sapphire as “Tie-Saph.” I have helpfully included a pronunciation guide for the aspiring laser physicist or forensic linguist, with the unambiguously correct pronunciations of each.
Theodore Maiman invented very first laser in 1960—a Chromium-doped Ruby crystal pumped by a flashlamp. Ruby lasers have since fallen out of fashion in favor of better gain media, like Nd:YAG8 or Ti:Sapphire. Nd:YAG and Ti:Sapphire are the short-pulse classics. Both lase in the near-IR, at 1064 nm and ~800 nm, respectively.9
Ti:Sapphire lasers are an absolute pain to tune up in practice, but still the go-to choice for single-digit femtosecond oscillators because of its ridiculously broad gain bandwidth.
Ytterbium-doped Crystals, usually Yb:YAG/Yb:KGW are the modern default for most ultrafast near-IR laser systems, due to its super low quantum defect (very, very power efficient), and super simple energy level structure. They usually lase near 1030 nm, and when frequency-doubled, produce my favorite shade of green.

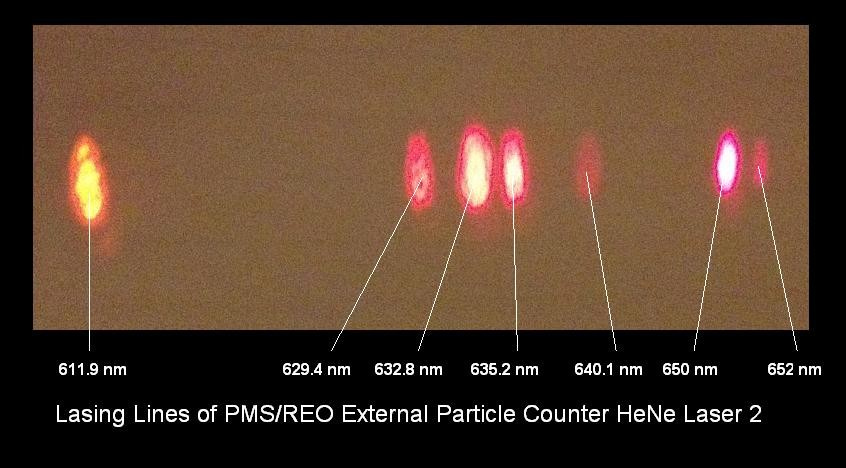
These use discrete vibrational or rotational energy levels of free gas molecules as the lasing medium. The CO₂ laser is the most representative in its class, the old workhorse of machining lasers. 10.6 um wavelength, usually. The other prototypical example is the Helium-Neon, or HeNe laser (usually 632 nm).10 It’s the experimentalist’s best friend, the beautiful bright red that aligns all your optics, with tubes that give you a really nasty shock if you don’t ground them properly. Ask me how I know.
Literally made of a liquid dye, though less used now on account of sometimes being extremely carcinogenic. The most common are Rhodamine (orange), Fluorescein (green), and Coumarin (blue). Coumarin comes from cinnamon and other plants, though I seriously doubt anyone has cold-pressed enough cinnamon to make a dye laser that way.
Properly a subtype of gas lasers, an excimer is an “excited dimer” but almost always lase in the ultraviolet, so form a different category in my head. The most famous are probably ArF & KrF, which set the Deep-UV 193 nm wavelength standard for photolithography in the 1970s and are still going. Also common in LASIK surgery.
A doped fiber optic, by far the most common type of laser now, for machining, telecom, whatever, because they’re so robust. Some common types are Erbium:Glass (the telecom standard 1550 nm amplifier), Nd:Glass, Yb:Glass, literally put any rare earth in a special type of glass (see for example ZBLAN fibers, one of the very few things that would be 10-100 times better if made in space).
The final category, less named for their gain medium than their architecture, are basically souped up LEDs. Exempli gratia:
Quantum Cascade Lasers
Vertical Cavity Surface-Emitting Lasers (VCSEL)
These are the Face ID Laser—if you look carefully at an iPhone in a dark room, you can see it flash!
Heterogeneous Integration Diodes with horrible acronyms like AlGaAsSb
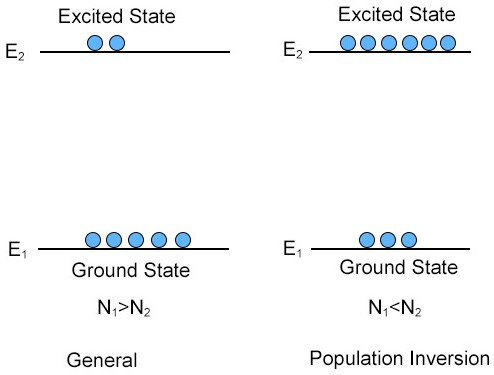
The stimulated absorption process requires a single excited electron. But gain requires many electrons in the excited state, and the pump provides the energy for it.
The goal of pumping a gain medium is to achieve population inversion,11 where you have more excited state electrons than ground state electrons, and you get net amplification of incoming photons.
There’s a bunch of ways to do it—we just learned one of them, you excite an electron with a photon of energy 𝜖, and it’ll jump up to an excited state with energy 𝜖.
There’s some nuances to this, though.
What do you think happens if you have a two level system, and you shine a continuous stream of energy-matched photons on it? Can you achieve population inversion like this? Think about it for a minute before scrolling down.
I’ll give you a hint, one of Einstein’s contributions in that seminal 1917 paper was showing that the absorption and stimulated emission probabilities are equal for any atomic transition, and they both involve a single incident photon…
The answer is: No, you cannot achieve steady-state population inversion with a two-level system.
Why? As your incoming photons get absorbed by the ground state electrons, the excited state gets more and more populated, and the reverse process, stimulated emission, gets more and more probable. Since the absorption and emission probabilities are equal, this process asymptotes to 50% excited, 50% ground state occupancy, with no net photon gain.
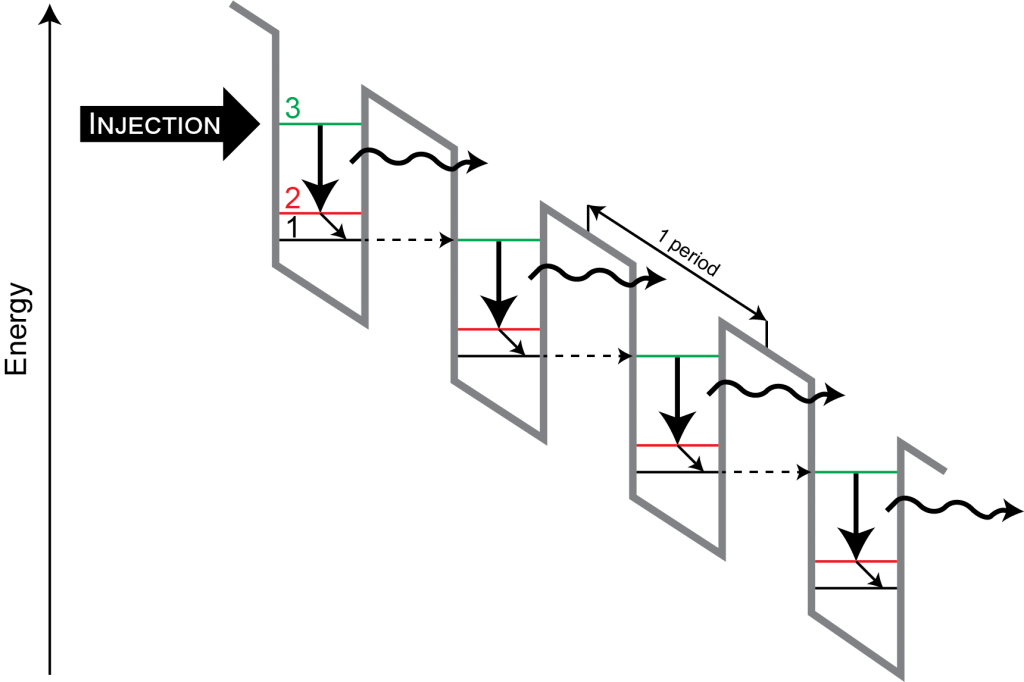
But you can, of course, cheat and use a three or four-level system, pumping on one transition and lasing on another. This is easier to explain in pictures than in words, so look at the figure below for a typical example of three and four level systems.
Usually, you have a pump source that excites the electrons into an upper level with a very short lifetime. That upper-most state rapidly decays into a middle state (confusingly called the ‘upper state’), whose transition to the ground is the lasing transition. The excess energy is lost as heat.12
The pump source can be any source of light. Flash- or arc-lamps are common (or used to be), but since specifically targeting the pump transition with photons of exactly the right energy is most efficient, most modern lasers actually use other lasers as the pump! But wait, how do those get pumped? Infinite regress problem?
No, usually, the pump laser is a semiconductor diode laser, and to pump those, you just ram as much electrical current through the diode as you possibly can, and a lot of those “hot” electrons stay in the upper state.
The final piece of the laser, often missing from otherwise good explanations, is the cavity. Real materials have many states, not two. In fact, real materials can lase over a very wide spectrum of electronic transitions. For an example, see the gain bandwidth of a Ti:Sapphire crystal (did you say it right?) below. It’s huge!
That’s why you need the cavity. The cavity is a positive feedback loop that selects a particular lasing wavelength (or set thereof) from the gain bandwidth, and suppresses the others. Usually, the cavity is a set of mirrors.
How does it work? In brief, the electric field of a laser cavity must exactly replicate itself on every round trip. This provides positive feedback via constructive interference.
Let’s sketch this process through the below thin-disk laser cavity:

We start at the first mirror with some light field. It has a magnitude and phase.
The light travels along the length of cavity, through the gain medium and outcoupler, and the phase of the light field advances by 𝜙 as it propagates.13 If the total round-trip length of the cavity is L,14 then the phase advance is 𝜙 = 2π * L/λ, where λ is the laser wavelength.
The phase advance is essentially the total round-trip cavity length, measured in units of wavelength, L/λ. Thus, for the field to perfectly repeat itself, the cavity needs to be an integer number of wavelengths long.
𝜙 = 2π n —> L = nλ
So for example, you could choose between a 700 nm laser line and 800 nm by changing the length of the cavity to match.
Additionally, the gain in the cavity must balance the loss in the cavity (including the out-coupled light, any mirror losses, etc.) at the lasing wavelength.
Gain(λ) = Loss
You increase the gain by increasing pump power, until Gain = Loss and you hit threshold. That’s lasing.
If you have those three things: A gain medium to provide the stimulated emission, a pump to provide the energy source, and a cavity to provide the feedback, you have a true laser.
You can have lots of other things that look like lasers, but aren’t. An LED and a laser diode are very similar, but the LED lacks the cavity. And indeed, if you slap an LED in a cavity you get a (bad) laser.
Thus we return to the premise of this article, with a little more understanding. If you have an atomic system with a bunch of states, and you pump it sufficiently hard, and there’s some sort of feedback mechanism, it will probably lase. I’ve collected a bunch of my favorite examples below, please enjoy!
Some of the animals in the zoo stretch the strict definition of laser, so I’m including a “Spectral Purity” score, where 1 means “yeah, this is a laser only in the loosest sense,” and 4 means “this meets the most pedantic definition of laser.” I reserve the right to be inconsistent and heavily biased in my scoring.
Nuclear-Pumped X-ray Lasers were a part of the Star Wars, or Strategic Defense Initiative under President Ronald Reagan.
There’s actually two kinds, one that uses a nuclear reactor to generate high energy ions that pump a laser medium by various means.15 The main application discussed in the linked review is to shoot down asteroids, by evaporating part of their surface with the nuclear-pumped laser, and using the resulting gas/plasma jet of ablated asteroid material to alter their trajectory.
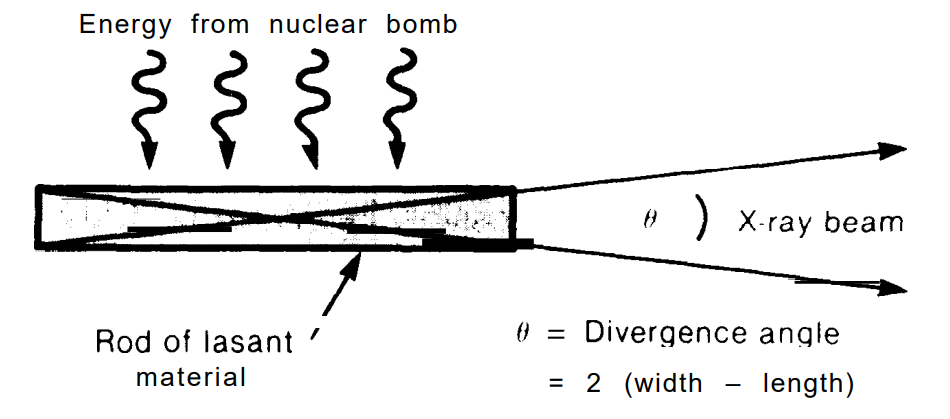
The other kind is more what you’d expect from a program called Star Wars, a nuclear bomb-pumped x-ray laser. There’s a really great Optica article on this, but apparently the results of the tests were only ever leaked, not published, and left observers “skeptical.”
The article claimed that the X-ray laser emitted peak power of several hundred terawatts at 1.4nm. Those figures have never been officially confirmed, but Livermore said in 1990 that the wavelength was the shortest ever emitted by a laser. The article did not identify the laser transition or test material, but Chapline told me the sample was “an organic pith material” from a weed growing on a vacant lot in Walnut Creek, Calif.
I love the banality of the origin story.
Congress had asked the weapons labs if they could do something with nuclear-weapon technology besides designing warheads, and the two decided that pumping an X-ray laser would be an interesting idea.16
Spectral Purity: 4 for reactor-pumped, 3 for bomb-pumped (it’s more an amplifier than a laser).
Cellular or biological lasers come in two flavors: random and engineered.
Random first: In bone tissue, the natural disorder of the hydroxyapatite mineral structure forms random optical cavities. When soaked in a fluorescent dye (e.g. Rhodamine), and pumped with a suitable source (e.g. a Ti:Sapphire), the various randomly oriented cavities will lase. It’s potentially useful, though this strikes me as more of an excuse to make bone lase than a useful technique:
Random cavities are strongly related to the structural properties of random media, [thus] the emission spectra can also be used to extract structural information of the samples.17
The engineered biolasers are only barely biological. You engineer a cell to express some fluorescent protein, and place it in an external optical cavity. The gain medium is engineered, the cavity isn’t biological, but it does lase and it does involve cells. It is, like random bone lasers, diagnostic of the cell. This technique also produces papers with extremely buzzword-dense titles guaranteed to excite a professor, e.g. Antigen-independent single-cell circulating tumor cell detection using deep-learning-assisted biolasers.
The random biolasers get a Spectral Purity score of 3/4 for the random natural cavities, and the engineered biolasers a 1/4. They count, but only barely—you really can’t just pump a cell and expect it to lase.
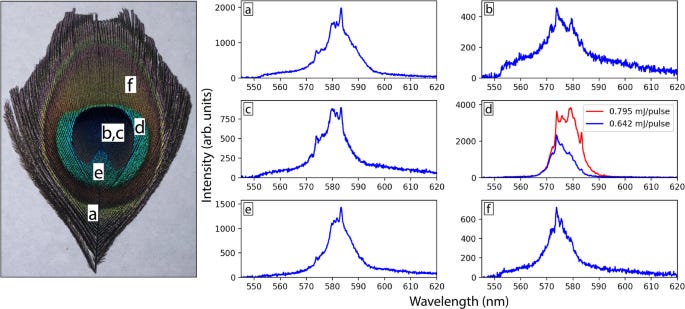
Both peacock feathers and butterfly wings are examples of natural photonic crystals, which are, in essence, extremely general tunable mirrors.18
So, someone had the bright idea to dip some feathers in rhodamine, pump it with 532 nm light, and it did indeed lase. Much to the authors’ surprise, and my own disappointment, the extremely cool natural photonic crystals were not responsible for the lasing. Nevertheless, they made a peacock feather lase. I expect future work from the authors to try again on the whole peacock.
Spectral Purity Score: 3/4. They had to dope it with a real laser gain medium, but the cavity was natural.
Theodor Hänsch and Arthur Schawlow wanted to make Jell-O lase. Their first twelve attempts with commercial Jell-O were unsuccessful—the food dyes were too absorptive. But they eventually succeeded by doping unflavored Knox-branded gelatin with rhodamine, fluorescein, umbelliferone (?), and coumarin. Each of these pseud-O-Jell-Os lased when pumped by a nitrogen laser.
Schawlow’s sense of humor comes through in the paper—he has two great notes.
“Since fluorescein is nearly nontoxic, this may well be the first edible laser material.”
If the pump beam remained stationary on the Jell-O, lasing faded after about 100 shots from local bleaching, but jiggling the Jell-O restored it.19
Sadly for Schawlow’s purity score, they had to make their own special Jell-O with strong fluorescent dyes. They also didn’t use a cavity, but the purpose was to demonstrate the gain medium, and the edges of the Jell-O are reflective enough to make a weak cavity, and I like Arthur Schawlow, so I’m giving this a 3/4.
A more modern version from Anwar et al.20 does a much more systematic job of looking into edible lasers. They explore combinations of edible microcavities made from oil droplets or silver leaf, edible fluorescent dyes, combine them, and put them in peach compote.
One of the applications they propose is really innovative, using multiple distinct laser lines as edible bar codes for fingerprinting food.
Spectral Purity for Anwar et al: 4/4, best example on this list, albeit not naturally formed.
This one sent me on a bit of a journey.
I’d heard the legends of Gin and Tonic lasers in grad school (and I love a good Gin and Tonic), but I couldn’t find any solid references in journals. What kind of scientist does this and doesn’t publish it?
I was able to find some references to a book by Stephen Wilk, reviewed here, who “traces the story to an experiment at Eastman Kodak labs, where two researchers found that very fast flashlamps could indeed excite blue laser pulses from an unidentified brand of tonic water.” But this experiment had no gin! So, I turned to Claude Deep Research.
The gin and tonic laser legend is actually conflating two experiments, the Eastman Kodak labs experiment above, and a paper from NIST’s predecessor the National Bureau of Standards, available here.21
The paper is so short I’ll just copy it in full. Read the last line.
Spectral Purity Score(s): 2/4. Lies! And a gaping hole in the literature…
The Martian Atmosphere is a natural CO₂ laser.
The effect was first definitively proved in 198122, by measuring the emission spectrum and showing it’s about 10^9 times brighter on the core CO₂ line than expected. They get a tiny, 10^-3 gain, and it’s single-pass, so not technically a laser, but a laser amplifier, and not a very good one.
However, another paper (Soviet this time) find that Mars’ atmosphere must lase given a certain temperature profile through the atmosphere, then infer from temperature measurements that not only is Mars always lasing on the daytime side, but Venus is too, but only when “internal gravity waves” (?) perturb the atmosphere.23 They estimate a gain of 2% through the Martian atmosphere, way above the 10^-3 of the earlier paper. Actually, with a decent cavity, that’s not bad for a natural laser—the single-pass gain for an artificial CO₂ laser is of order 0.2% per cm.
The Ant Nebula is apparently also lasing, which is somehow evidence of a double star system, because otherwise the conditions in the gases aren’t right, something something. Astronomers have absolutely crazy chains of evidence. Off-topic, but my favorite example of astronomer bootstrap reasoning is Terrence Tao’s talk on The Cosmic Distance Ladder. Fantastic talk on how we learned to measure distances between stars.
Spectral Purity Score: 3/4, a true laser amplifier in want of a cavity.
To wrap up this article, it is sadly necessary to mention that Schawlow is not quite right. Alas, some things simply will not lase, and there’s a general principle involved. Several actually, but I’ll touch on two.
Gain is linear with pumping strength. Pump power → population inversion → gain coefficient. So, if you have some sort of lossy effect that is superlinear with pump power (Auger recombination, for example, where the energy just bleeds off uselessly as heat), the thing won’t lase. Surprisingly, silicon is one of these materials, despite its ubiquity.
There’s another, more fundamental constraint too. The upper-state lifetime, that is, the amount of time an electron will stay excited before spontaneously emitting a photon and dropping down, is proportional to 1 / f^3, where f is the lasing frequency:
\(\tau \propto \frac{1}{f^3}\)
Thus, the pump power, to sustain inversion of N electrons in the upper state, is in dynamic balance, and scales like the total energy of pump photons over upper-state lifetime
\(P_{pump}\propto N E_{ph} / \tau \propto f^4, \text{ recalling } E_{ph} = hf\)
This single fact explains why near-IR lasers require milliwatts, visible require watts, UV require kilowatts, and X-ray lasers require nuclear weapons. This is a major impediment towards the holy grail—the gamma ray laser (graser).24
And so there we have it. A surprising number of things will lase if pumped… but for silicon, and gamma ray lasers, you really can’t hit them hard enough, or at least not yet.